Chapter 15: Q11. (page 570)
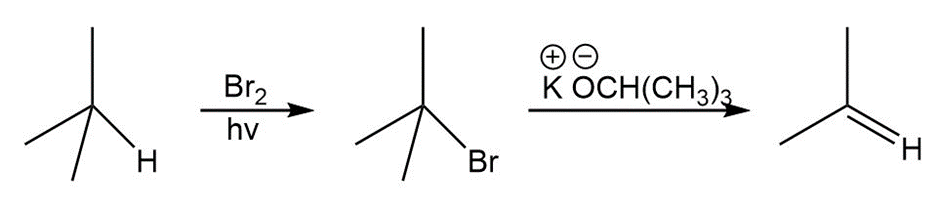
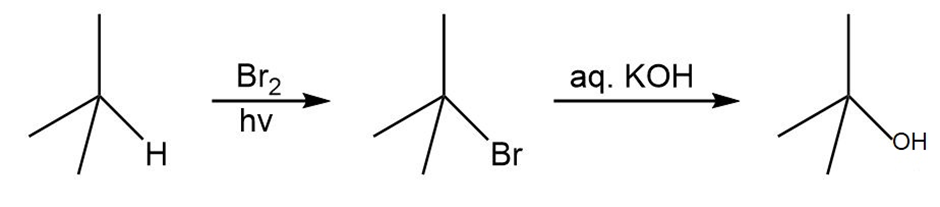
Question: Synthesize each compound from (CH3)3 CH.
a.
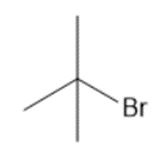
b.
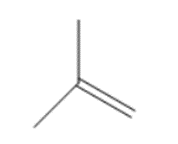
c.
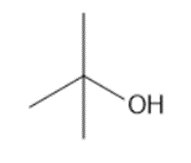
Short Answer
Answer
a.
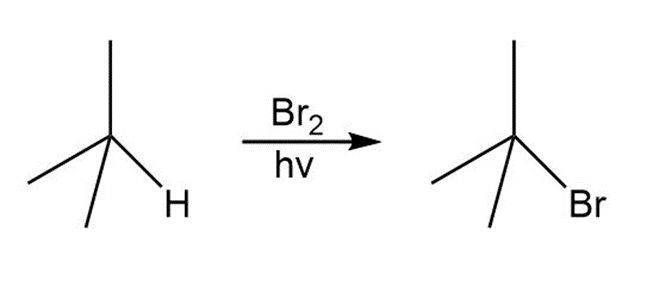
b.
c.
Learning Materials
Features
Discover
Chapter 15: Q11. (page 570)
Question: Synthesize each compound from (CH3)3 CH.
a.

b.

c.

Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question. Devise a synthesis of hexane-2,3-diol from propane as the only source of carbon atoms. You may use any other required organic or inorganic reagents.
Question: As we will learn in Chapter 30, styrene derivatives such as A can be polymerized by way of cationic rather than radical intermediates. Cationic polymerization is an example of electrophilic addition to an alkene involving carbocations.
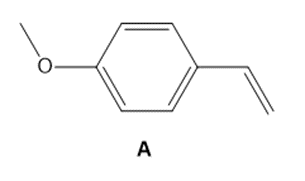
a. Draw a short segment of the polymer formed by the polymerization of A.
b. Why does A react faster than styrene (C6H5CH=CH2)in a cationic polymerization?
Question: Draw the product(s) formed when each alkene is treated with either [1] HBr alone; or [2] HBr in the presence of peroxides.
a.
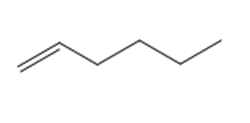
b.
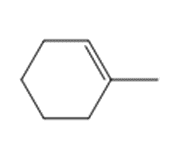
c.
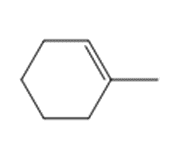
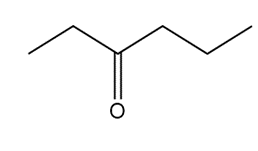
Question: Devise a synthesis of each compound using CH3CH3 as the only source of carbon atoms. You may use any other required organic or inorganic reagents.
a.
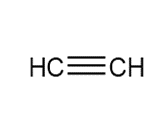
b.
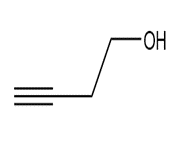
c.
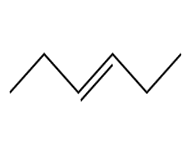
d.
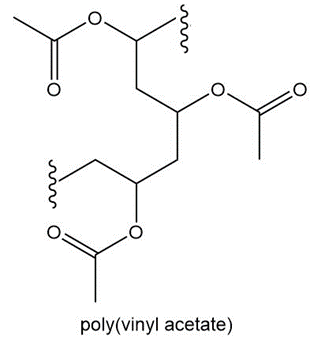
Question: (a)Draw the structure of polystyrene, the chapter-opening molecule, which is formed by polymerizing the monomer styrene, C6H5CH=CH2. (b) What monomer is used to form poly(vinyl acetate), a polymer used in paints and adhesives?
What do you think about this solution?
We value your feedback to improve our textbook solutions.