Chapter 14: Q21P (page 527)
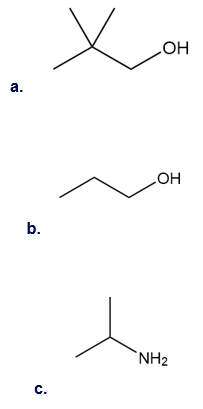
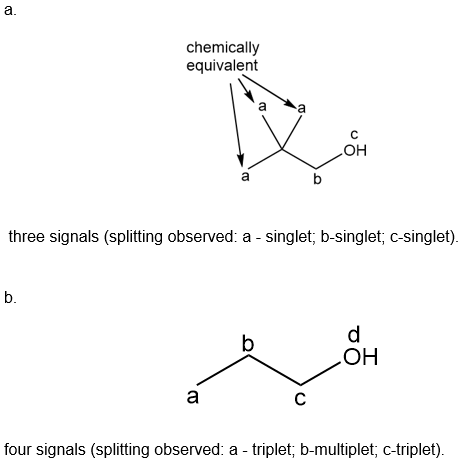
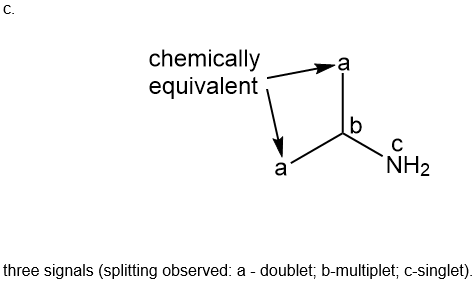
Question: How many signals are present in the \({}^{\bf{1}}{\bf{H}}\)NMR spectrum for each molecule? What splitting is observed in each signal?
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q21P (page 527)
Question: How many signals are present in the \({}^{\bf{1}}{\bf{H}}\)NMR spectrum for each molecule? What splitting is observed in each signal?



All the tools & learning materials you need for study success - in one app.
Get started for free
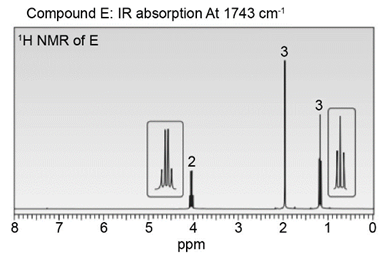
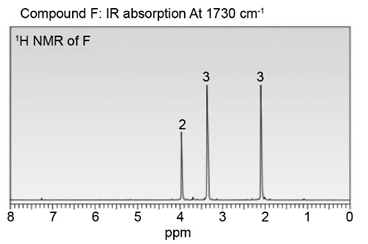
Question: Identify the structures of isomers E and F (molecular formula C4H802 ). Relative areas are given above each signal.
a.
b.
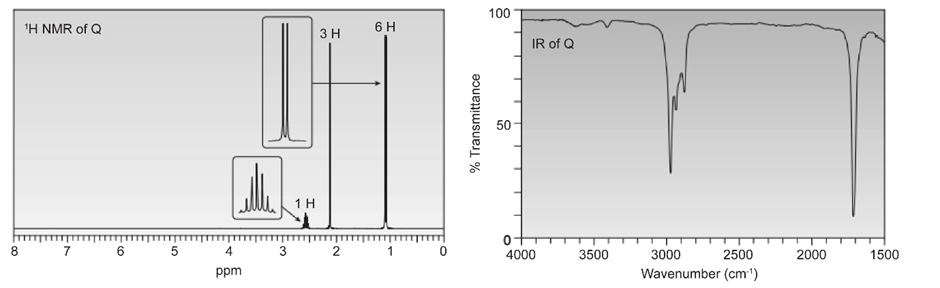
Question. Compound Q has molecular formula C5H9CIO2. Deduce the structure of P from its 1H and 13C -NMR spectra.
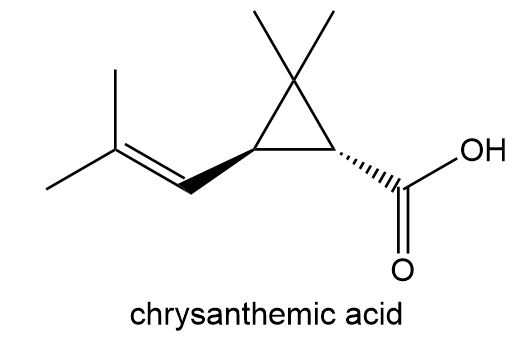
Esters of chrysanthemic acid are naturally occurring insecticides. How many lines are present in the NMR spectrum of chrysanthemic acid?
Identify products A and B from the given \({}^{\bf{1}}{\bf{H}}\) NMR data.
(A) Treatment of \({\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{ = CHCOC}}{{\bf{H}}_{\bf{3}}}\) with one equivalent of HCl forms compound A. A exhibits the following absorptions in its \({}^{\bf{1}}{\bf{H}}\)NMR spectrum: 2.2 (singlet, 3 H), 3.05 (triplet, 2 H), and 3.6 (triplet, 2H) ppm. What is the structure of A?
(B) Treatment of acetone \(\left( {{{\left( {{\bf{C}}{{\bf{H}}_{\bf{3}}}} \right)}_{\bf{2}}}{\bf{C = O}}} \right(\)with dilute aqueous base forms B. Compound B exhibits four singlets in its \({}^{\bf{1}}{\bf{H}}\) NMR spectrum at 1.3 (6 H), 2.2 (3 H), 2.5 (2 H), and 3.8 (1 H) ppm. What is the structure of B?
What do you think about this solution?
We value your feedback to improve our textbook solutions.