Chapter 14: Q.21558-14-59P (page 565)
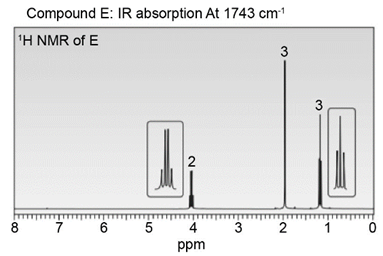
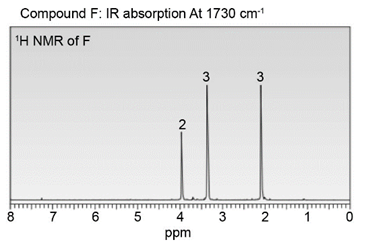
Question: Identify the structures of isomers E and F (molecular formula C4H802 ). Relative areas are given above each signal.
a.
b.
Short Answer
Answer
a.
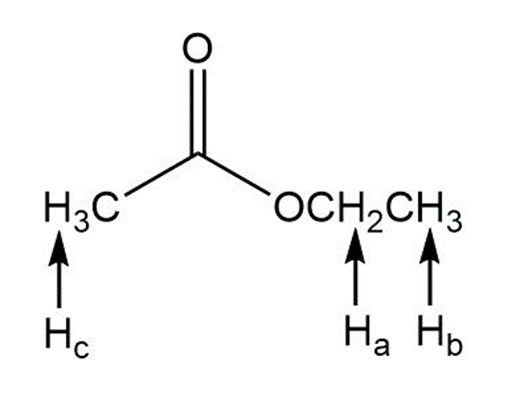
b.
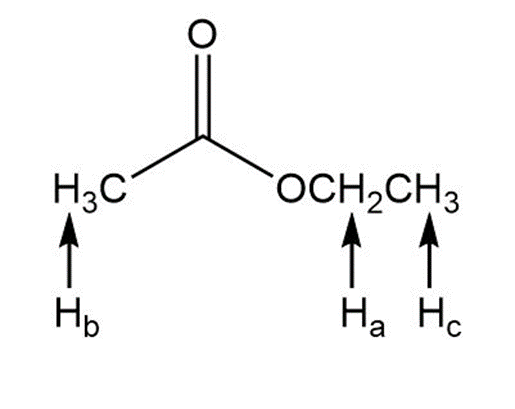
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-59P (page 565)
Question: Identify the structures of isomers E and F (molecular formula C4H802 ). Relative areas are given above each signal.
a.

b.

Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: The \(^{\bf{1}}{\bf{H}}\) NMR spectrum of \({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{OH}}\) recorded on a 500 MHz NMR spectrometer consists of two signals, one due to the \({\bf{C}}{{\bf{H}}_{\bf{3}}}\) protons at 1715 Hz and one due to the OH proton at 1830 Hz, both measured downfield from TMS. (a) Calculate the chemical shift of each absorption. (b) Do the \({\bf{C}}{{\bf{H}}_{\bf{3}}}\) protons absorb upfield or downfield from the OH proton?
Question:Propose a structure consistent with each set of spectral data:
a. C4H8Br2: IR peak at 3000–2850cm-1 ;
NMR (ppm):
1.87 (singlet, 6 H)
3.86 (singlet, 2 H)
b. C3H6Br2: IR peak at 3000–2850cm-1 ;
NMR (ppm):
2.4 (quintet)
3.5 (triplet)
c. C5H10O2: IR peak at 1740cm-1 ;
NMR (ppm):
1.15 (triplet, 3 H) 2.30 (quartet, 2 H)
1.25 (triplet, 3 H) 4.72 (quartet, 2 H)
d . C6H14O: IR peak at 3600-3200 cm-1 ;
NMR (ppm):
0.8 (triplet, 6 H) 1.5 (quartet, 4 H)
1.0 (singlet, 3 H) 1.6 (singlet, 1 H)
e. C6H14O: IR peak at 3000-2850cm-1 ;
NMR (ppm):
1.10 (doublet, relative area = 6)
3.60 (septet, relative area = 1)
f . C3H6O: IR peak at 1730cm-1 ;
NMR (ppm):
1.11 (triplet)
2.46 (multiplet)
9.79 (triplet)
Identify A and B, isomers of molecular formula C3H4Cl2 , from the given 1HNMR data: Compound A exhibits signals at 1.75 (doublet, 3H,J = 6.9 Hz) and 5.89 (quartet, 1H, J = 6.9 Hz) ppm. Compound B exhibits signals at 4.16 (singlet, 2 H), 5.42 (doublet, 1H, J = 1.9 Hz), and 5.59 (doublet, 1H, J = 1.9 Hz) ppm.
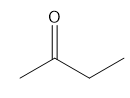
Question: For each compound, first label each different type of proton and then rank the protons in order of increasing chemical shift.
a.
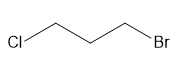
b.
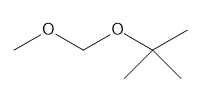
c.
Question: Reaction of C6H5CH2CH2OH with CH3COCl affords compound W, which has molecular formula C10H12O2. W shows prominent IR absorptions at 3088–2897, 1740, and 1606cm-1 . W exhibits the following signals in its 1 H NMR spectrum: 2.02 (singlet), 2.91 (triplet), 4.25 (triplet), and 7.20–7.35 (multiplet) ppm. What is the structure of W? We will learn about this reaction in Chapter 22.
What do you think about this solution?
We value your feedback to improve our textbook solutions.