Chapter 14: Q.21558-14-29P. (page 558)
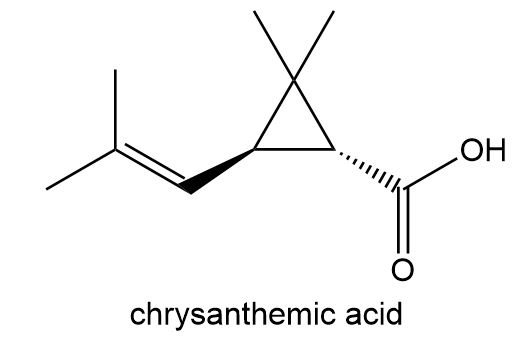
Esters of chrysanthemic acid are naturally occurring insecticides. How many lines are present in the NMR spectrum of chrysanthemic acid?
Short Answer
The 13C NMR spectrum consists of nine lines.
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-29P. (page 558)
Esters of chrysanthemic acid are naturally occurring insecticides. How many lines are present in the NMR spectrum of chrysanthemic acid?

The 13C NMR spectrum consists of nine lines.
All the tools & learning materials you need for study success - in one app.
Get started for free
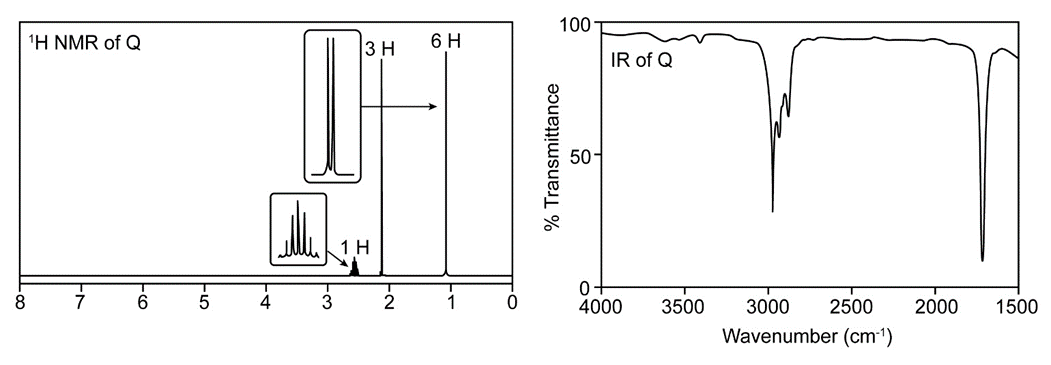
Question. Treatment of butan-2-one (CH3COCH2CH3) with a strong base followed by CH3l forms a compound Q, which gives a molecular ion in its mass spectrum at 86. The IR (> 1500 only) and 1H-NMR spectrum of Q is given below. What is the structure of Q?
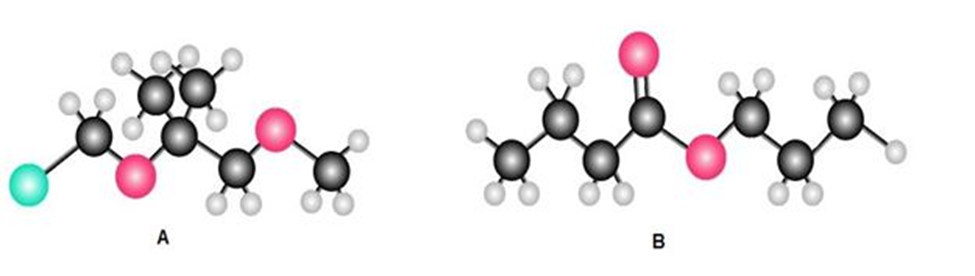
Question: (a) How many 1H NMR signals does each of the following compounds exhibit? (b) How many 13C NMR signals does each compound exhibit?
Rank each group of protons in order of increasing chemical shift.
a.
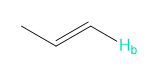
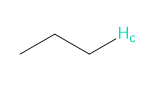
b.
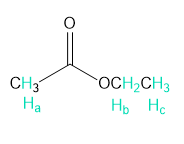
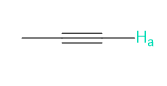
Into how many peaks will each proton shown in green be split?
a.
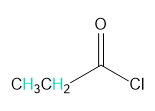
b.
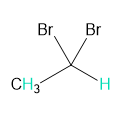
c.
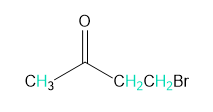
d.
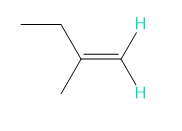
e.
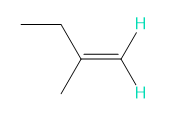
f.
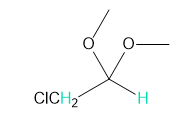
For each compound, which of the protons on the highlighted carbons absorbs farther downfield?
a.
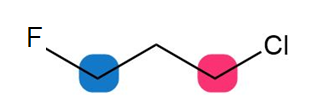
b.
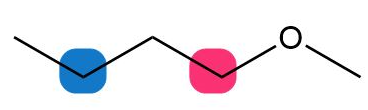
c.
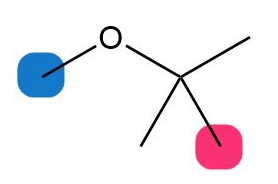
What do you think about this solution?
We value your feedback to improve our textbook solutions.