Chapter 14: Q.21558-14-14P. (page 546)
Into how many peaks will each proton shown in green be split?
a.
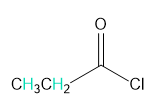
b.
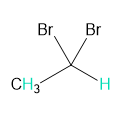
c.
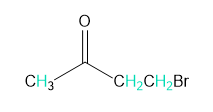
d.
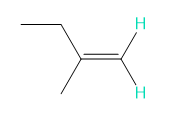
e.
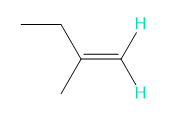
f.
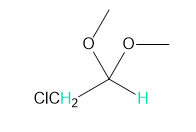
Short Answer
a. 3 and 4
b. 2 and 4
c. 1, 3, and 3
d. 2 and 2
e. 2 and 2
f. 2 and 3
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-14P. (page 546)
Into how many peaks will each proton shown in green be split?
a.

b.

c.

d.

e.

f.

a. 3 and 4
b. 2 and 4
c. 1, 3, and 3
d. 2 and 2
e. 2 and 2
f. 2 and 3
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Identify the structures of isomers H and I (molecular form C8H11N)
a.
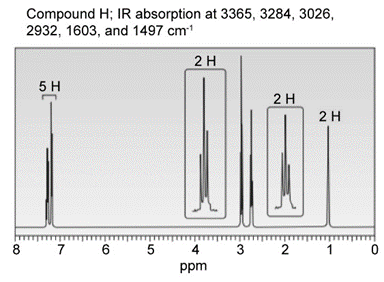
b.
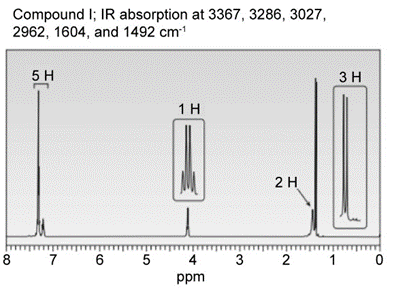
Question: Cyclohex-2-enone has two protons on its carbon-carbon double bond (labeled Ha and Hb ) and two protons on the carbon adjacent to the double bond (labeled Hc ). (a) If Jab = 11 Hz and Jbc = 4 Hz, sketch the splitting pattern observed for each proton on the hybridized carbons. (b) Despite the fact that Ha is located adjacent to an electron-withdrawing C=O, its absorption occurs up-field from the signal due to Hb(6.0 vs. 7.0 ppm). Offer an explanation.
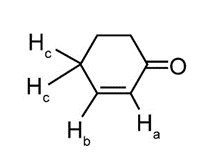
Question: What splitting pattern is observed for each proton in the following compounds?
a.
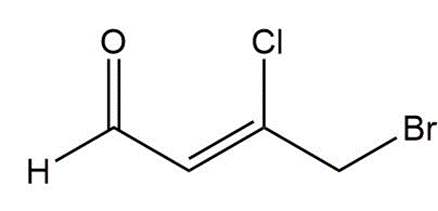
b.
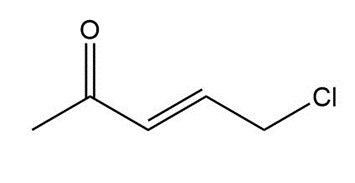
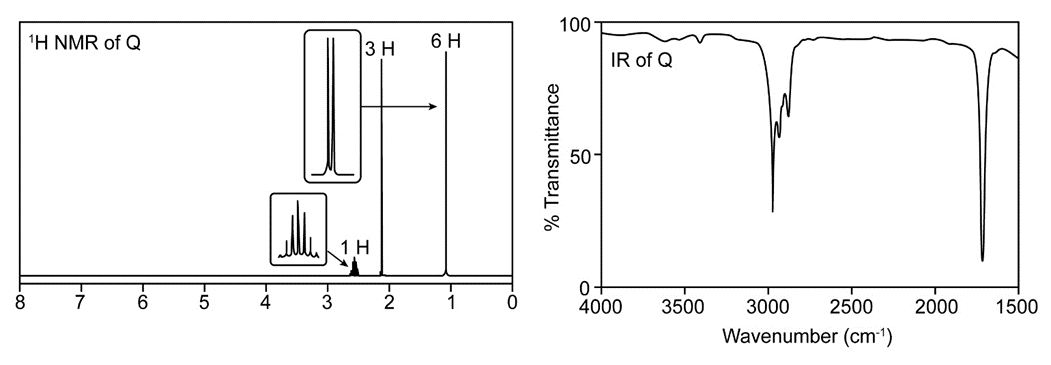
Question. Treatment of butan-2-one (CH3COCH2CH3) with a strong base followed by CH3l forms a compound Q, which gives a molecular ion in its mass spectrum at 86. The IR (> 1500 only) and 1H-NMR spectrum of Q is given below. What is the structure of Q?
Question: For each compound, which of the protons on the highlighted carbons absorbs farther downfield?
a. 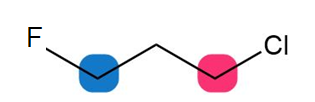
b. 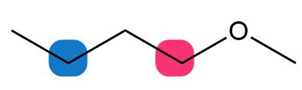
c. 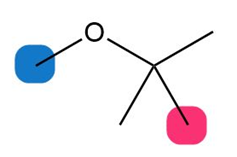
What do you think about this solution?
We value your feedback to improve our textbook solutions.