Chapter 14: Q.21558-14-17P. (page 549)
How many peaks are present in the NMR signal of each labelled proton?
a.
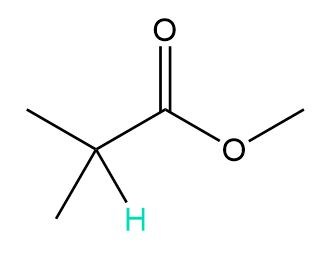
b.
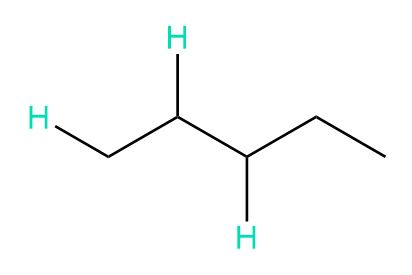
c.
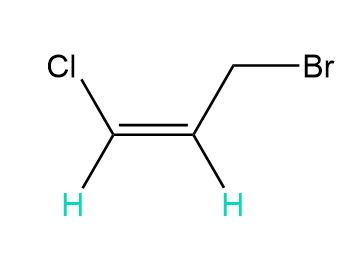
d.
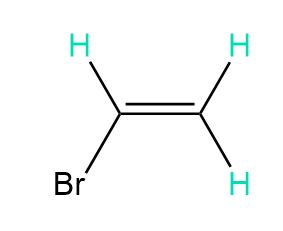
Short Answer
a. 7 peaks
b. 3, 5, 12, and 6 peaks
c. 2 and 6 peaks
d. 4, 4, and 4 peaks
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-17P. (page 549)
How many peaks are present in the NMR signal of each labelled proton?
a.

b.

c.

d.

a. 7 peaks
b. 3, 5, 12, and 6 peaks
c. 2 and 6 peaks
d. 4, 4, and 4 peaks
All the tools & learning materials you need for study success - in one app.
Get started for free
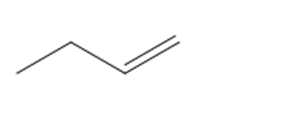
How many lines are observed in the \({}^{{\bf{13}}}{\bf{C}}\) NMR spectrum of each compound?
a.
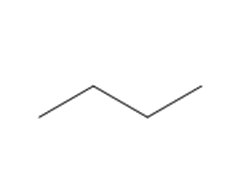
b.
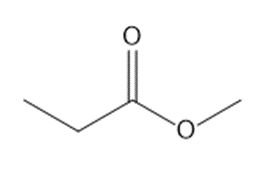
c.
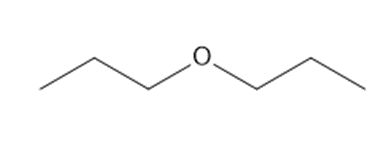
d.
Question: Identify the carbon atoms that give rise to the signals in the 13C NMR spectrum of each compound.
a. CH3CH2CH2CH2OH ; 13CNMR: 14, 19, 35, and 62 ppm
b. (CH3)2CHCHO ; 13C NMR: 16, 41, and 205 ppm
c. CH2=CHCHOHCH3 ; 13C NMR: 23, 69, 113, and 143 ppm
Question. Reaction of aldehyde D with amino alcohol E in the presence of NaH forms F (molecular formula C11H15NO2). F absorbs at 1730 cm-1in its IR spectrum. F also shows eight lines in its 13C-NMR spectrum, and gives the following -NMR spectrum: 2.32 (singlet, 6 H), 3.05 (triplet, 2 H), 4.20 (triplet, 2 H), 6.97 (doublet, 2 H), 7.82 (doublet, 2 H), and 9.97 (singlet, 1 H) ppm. Propose a structure for F. We will learn about this reaction in Chapter 18.
How many signals are present in the 1HNMR spectrum for each molecule? What splitting is observed in each signal?
a.
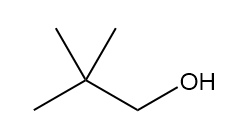
b.
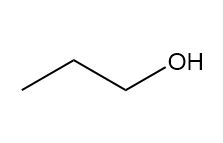
c.
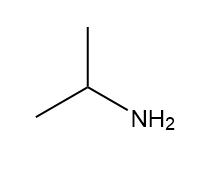
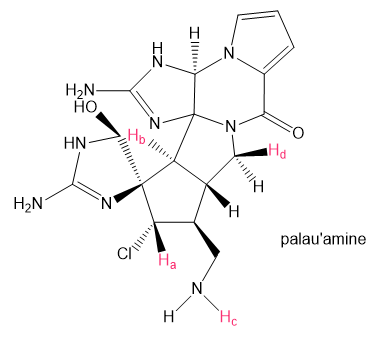
How many peaks are observed in the 1HNMR signal for each proton shown in red in palau'amine, the complex chapter-opening molecule?
What do you think about this solution?
We value your feedback to improve our textbook solutions.