Chapter 14: Q.21558-14-16P. (page 546)
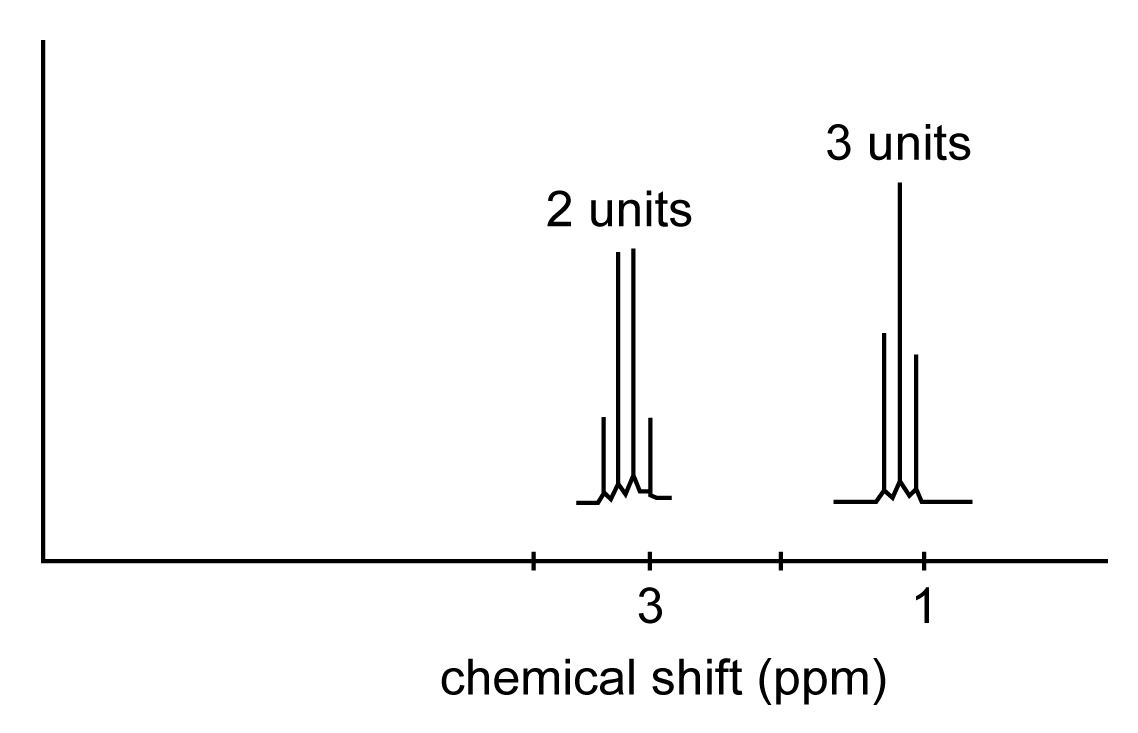
Sketch the NMR spectrum of CH3CH2Cl , giving the approximate location of each NMR signal.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-16P. (page 546)
Sketch the NMR spectrum of CH3CH2Cl , giving the approximate location of each NMR signal.

All the tools & learning materials you need for study success - in one app.
Get started for free
Label each statement as True or False.
a. When a nucleus is strongly shielded, the effective field is larger than the applied field and the absorption shifts downfield.
b. When a nucleus is strongly shielded, the effective field is smaller than the applied field and the absorption is shifted upfield.
c. A nucleus that is strongly deshielded requires a lower field strength for resonance.
d. A nucleus that is strongly shielded absorbs at a larger δ value.
Question: As we will learn in Chapter 20, reaction of (CH3)2CO with followed by affords compound D, which has a molecular ion in its mass spectrum at 84 and prominent absorptions in its IR spectrum at 3600–3200, 3303, 2938, and 2120cm-1 . D shows the following 1 H NMR spectral data: 1.53 (singlet, 6 H), 2.37 (singlet, 1 H), and 2.43 (singlet, 1 H) ppm. What is the structure of D?
Question: How many different types of protons are present in each compound?
Question: Treatment of 2-methylpropanenitrile [(CH3)2CHCN] with CH3CH2CH2MgBr, followed by aqueous acid, affords compound V, which has molecular formula C7H14O. V has a strong absorption in its IR spectrum at 1713 cm-1, and gives the following 1 H NMR data: 0.91 (triplet, 3 H), 1.09 (doublet, 6 H), 1.6 (multiplet, 2 H), 2.43 (triplet, 2 H), and 2.60 (septet, 1 H) ppm. What is the structure of V? We will learn about this reaction in Chapter 22.
What do you think about this solution?
We value your feedback to improve our textbook solutions.