Chapter 19: Q39 (page 758)
Rank the compounds in each group in order of increasing basicity.
a.
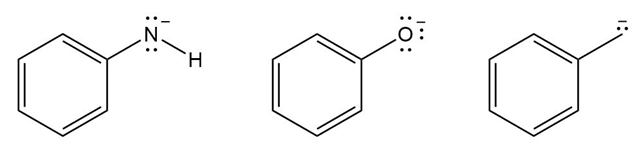
b.

Short Answer
a.
Order of basicity: 3>1>2
b.
Order of basicity:
Learning Materials
Features
Discover
Chapter 19: Q39 (page 758)
Rank the compounds in each group in order of increasing basicity.
a.

b.

a.
Order of basicity: 3>1>2
b.
Order of basicity:
All the tools & learning materials you need for study success - in one app.
Get started for free
Which carboxylic acid has the lower , pyruvic acid or acetoacetic acid ? Explain your choice.
Propose a structure for D (molecular formula ) consistent with the given spectroscopic data.
NMR signals at 30,36,128,133, 139, and 179 ppm
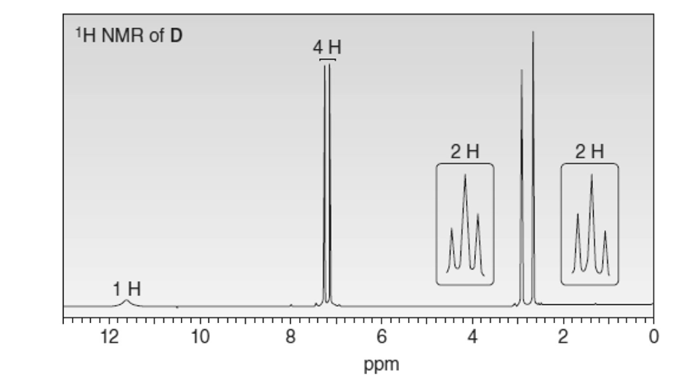
Match the NMR data to the appropriate structure.
Spectrum [1]: signals at 14, 22, 27, 34, 181 ppm
Spectrum [2]: signals at 27, 39, 186 ppm
Spectrum [3]: signals at 22, 26, 43, 180 ppm
a.
b.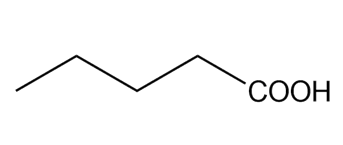
c.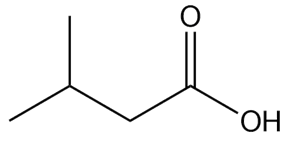
Question: How many tetrahedral stereogenic centers does contain? Draw its enantiomer. How many of its double bonds can exhibit cis-trans isomerism? Considering both its double bonds and its tetrahedral stereogenic centers, how many stereoisomers are possible for ?
Calculate the isoelectric point for each amino acid.
a. cysteine: (COOH) = 2.05; =10.25
b. methionine: (COOH) = 2.28; =9.21
What do you think about this solution?
We value your feedback to improve our textbook solutions.