Chapter 19: Q42 (page 759)
Which carboxylic acid has the lower , pyruvic acid or acetoacetic acid ? Explain your choice.
Short Answer
Pyruvic acid is the carboxylic acid that has a lower value.
Learning Materials
Features
Discover
Chapter 19: Q42 (page 759)
Which carboxylic acid has the lower , pyruvic acid or acetoacetic acid ? Explain your choice.
Pyruvic acid is the carboxylic acid that has a lower value.
All the tools & learning materials you need for study success - in one app.
Get started for free
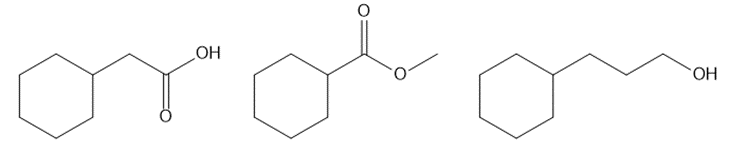
Question: Rank the following compounds in order of increasing boiling point. Which compound is the most water soluble? Which compound is the least water soluble?
Question: Draw the structure corresponding to each name.
The of p-methylthiophenol is 9.53. Is p-methylthiophenol more or less reactive in electrophilic aromatic substitution than phenol?
For each amino acid , draw its neutral, positively charged, and negatively charged forms. Which form predominates at pH = 1, 6, and 11? What is the structure of each amino acid at its isoelectric point?
a. methionine
b. serine
Because phenol is less acidic than a carboxylic acid, it can be deprotonated by NaOH but not by the weaker base . Using this information, write out an extraction sequence that can be used to separate , benzoic acid, and cyclohexanol. Show what compound is present in each layer at each stage of the process, and if it is present in its neutral or ionic form.
What do you think about this solution?
We value your feedback to improve our textbook solutions.