Chapter 19: Q38 (page 758)
Rank the compounds in each group in order of increasing acidity.
a.
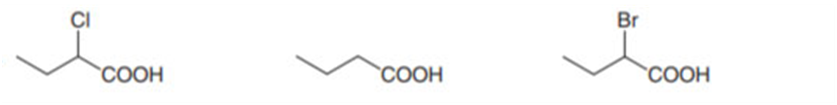
b.
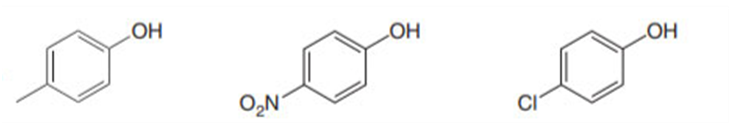
Short Answer
a.
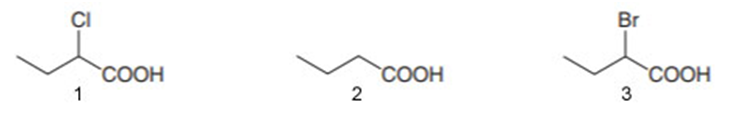
Order of acidity: 1>3>2
b.
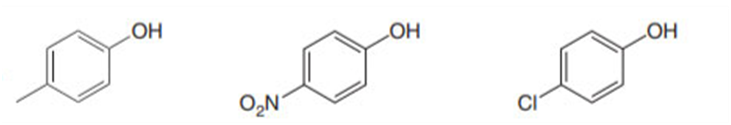
Order of acidity: 2> 3>1
Learning Materials
Features
Discover
Chapter 19: Q38 (page 758)
Rank the compounds in each group in order of increasing acidity.
a.

b.

a.

Order of acidity: 1>3>2
b.

Order of acidity: 2> 3>1
All the tools & learning materials you need for study success - in one app.
Get started for free
Identify X in the following equation, and explain how hexanoic acid (Section 19.2B) is formed by this stepwise reaction sequence.

Can octane and octan-1-ol be separated using an aqueous extraction procedure? Explain why or why not.
Question: The values for the carboxy and ammonium protons of phenylalanine are 2.58 and 9.24, respectively. What is the isoelectric point of phenylalanine? Draw the structure of phenylalanine at its isoelectric point.
Question: Give the IUPAC name for each metal salt of a carboxylate anion.
a. 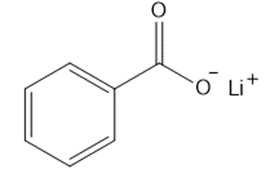
b. 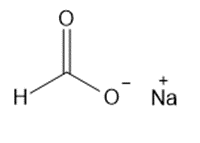
c. 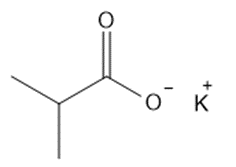
d. 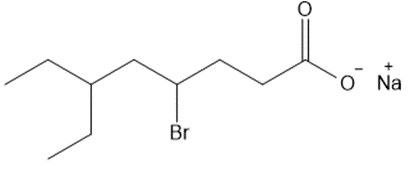
For each amino acid , draw its neutral, positively charged, and negatively charged forms. Which form predominates at pH = 1, 6, and 11? What is the structure of each amino acid at its isoelectric point?
a. methionine
b. serine
What do you think about this solution?
We value your feedback to improve our textbook solutions.