Chapter 19: Q17. (page 746)
Question: Rank the compounds in each group in order of increasing acidity. Rank the compounds in each group in order of increasing acidity.
a. 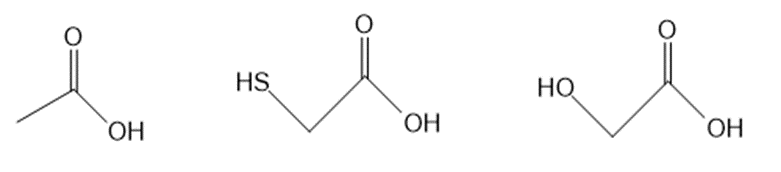
b. 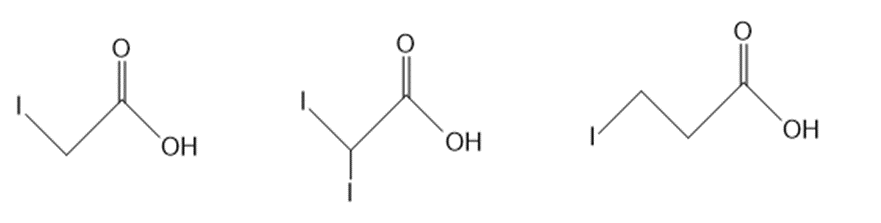
Short Answer
Answer
a.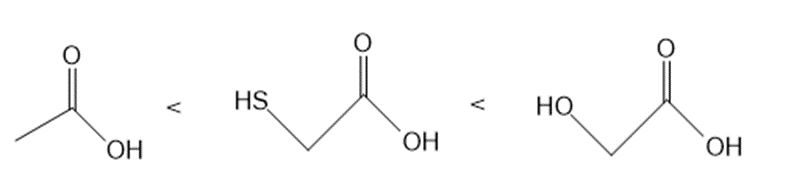
b.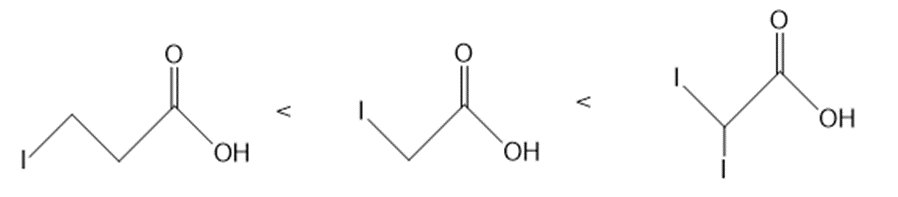
Learning Materials
Features
Discover
Chapter 19: Q17. (page 746)
Question: Rank the compounds in each group in order of increasing acidity. Rank the compounds in each group in order of increasing acidity.
a. 
b. 
Answer
a.
b.
All the tools & learning materials you need for study success - in one app.
Get started for free
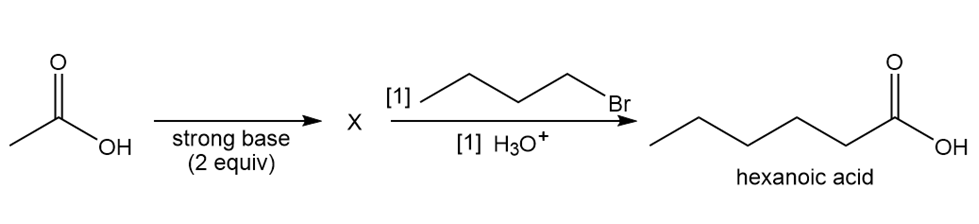
Identify X in the following equation, and explain how hexanoic acid (Section 19.2B) is formed by this stepwise reaction sequence.
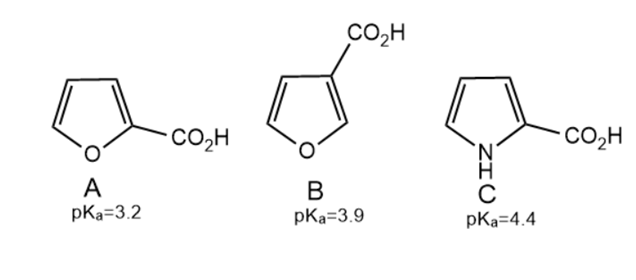
Explain why the of compound A is lower than the ’s of both compounds B and C.
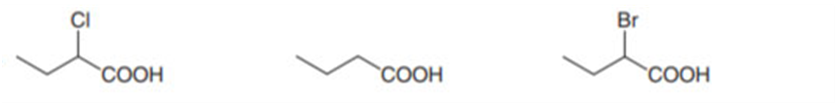
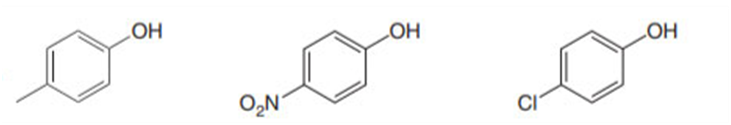
Rank the compounds in each group in order of increasing acidity.
a.
b.
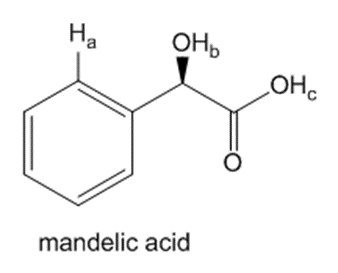
Question: Rank the labeled protons in mandelic acid, a naturally occurring carboxylic acid in plumsand peaches, in order of increasing acidity. Explain in detail why you chose this order.
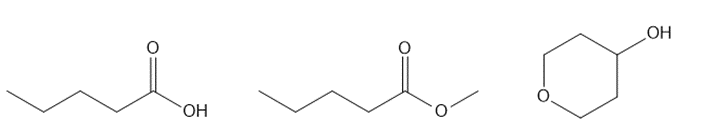
Question: Explain how you could use IR spectroscopy to distinguish among the following three compounds.
What do you think about this solution?
We value your feedback to improve our textbook solutions.