Chapter 19: Q16. (page 746)
Question: Match each of the following values (3.2, 4.9, and 0.2) to the appropriate carboxylic acid:
(a)
Short Answer
Answer
a. The value of is 4.9.
b. The value of is 3.2.
c. The value of is 3.2.
Learning Materials
Features
Discover
Chapter 19: Q16. (page 746)
Question: Match each of the following values (3.2, 4.9, and 0.2) to the appropriate carboxylic acid:
(a)
Answer
a. The value of is 4.9.
b. The value of is 3.2.
c. The value of is 3.2.
All the tools & learning materials you need for study success - in one app.
Get started for free
Proline is an unusual amino acid because its N atom on the α carbon is part of a five-membered ring
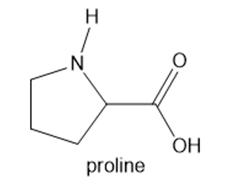
a. Draw both enantiomers of proline.
b. Draw proline in its zwitterionic form.
Question: Draw the positively charged, neutral, and negatively charged forms for the amino acid glycine.
Which species predominates at pH 11? Which species predominates at pH 1?
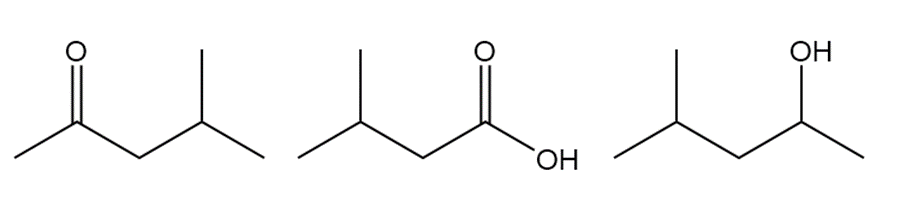
Question: Rank the following compounds in order of increasing boiling point.
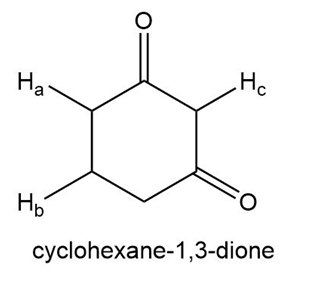
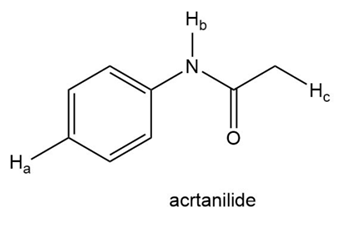
Draw all resonace strucutres of the conjugate bases formed by removal of the labeled protons in cyclohexane-1,3-dione and acetanilide. For each compound, rank these protons in order of increasing acidity and explain the order you chose.
a.
b.
Question: Two other commonly used sulfonic acids are methanesulfonic acid and trifluoromethanesulfonic acid . Which has the weaker conjugate base? Which conjugate base is the better leaving group? Which of these acids has the higher ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.