Chapter 19: Q18. (page 748)
Question: Rank the compounds in each group in order of increasing acidity.
a. 
b. 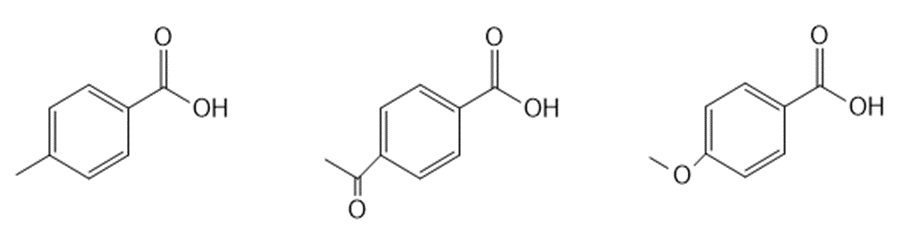
Short Answer
Answer
a.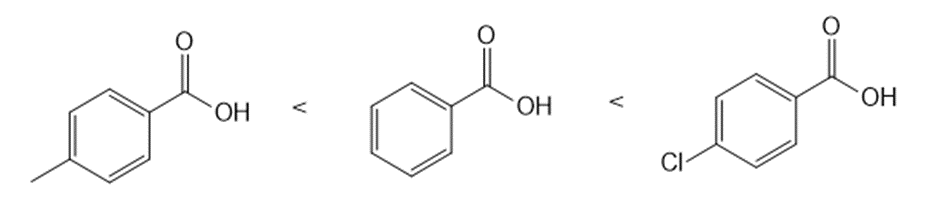
b.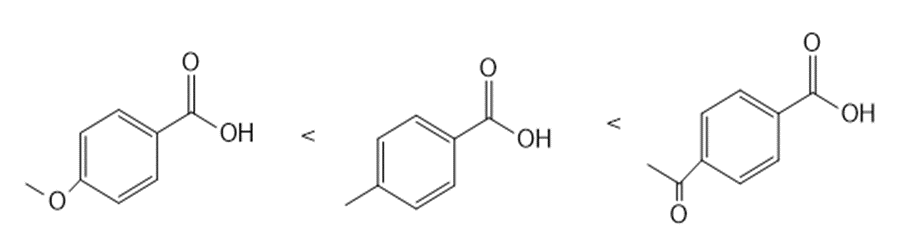
Learning Materials
Features
Discover
Chapter 19: Q18. (page 748)
Question: Rank the compounds in each group in order of increasing acidity.
a. 
b. 
Answer
a.
b.
All the tools & learning materials you need for study success - in one app.
Get started for free
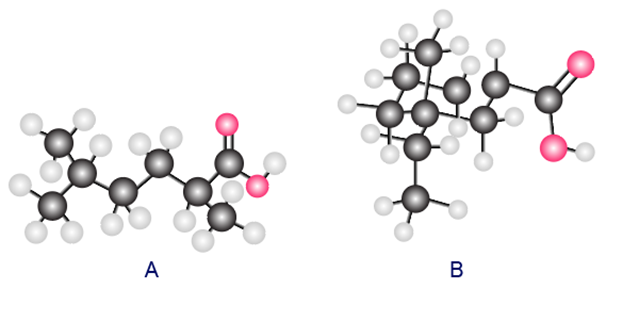
Question: Answer each question for A and B depicted in the ball-and-stick models.
Question: Draw the cation that results when a carboxylic acid is protonated on the oxygen of the OH group. Explain why protonation of the carbonyl oxygen is preferred to protonation of the hydroxy oxygen.
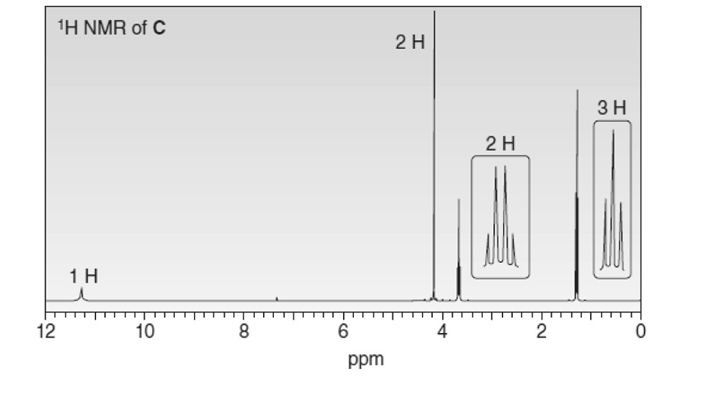
An unknown compound C (molecular formula ) exhibits IR absorption at 3600-2500 and 1734 localid="1649052784484" , as well as the following NMR spectrum. What is the structure of C?
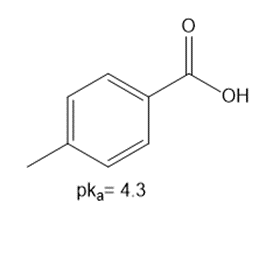
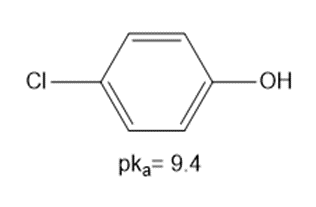
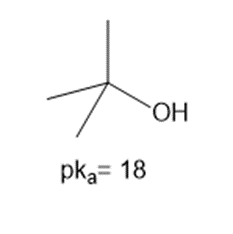
Question: Using the table in Appendix A, determine whether each of the following bases is strong enough to deprotonate the three compounds listed below. Bases: [1]
a.
b.
c.
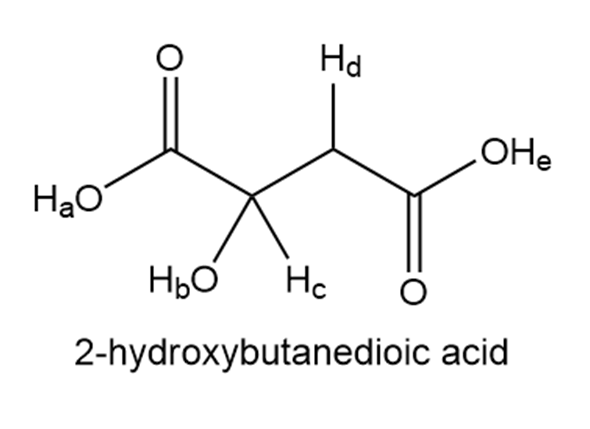
2-Hydroxybutanedioic acid occurs naturally in apples and other fruits. Rank the labeled protons in order of increasing acidity and explain in detail the order you chose.
What do you think about this solution?
We value your feedback to improve our textbook solutions.