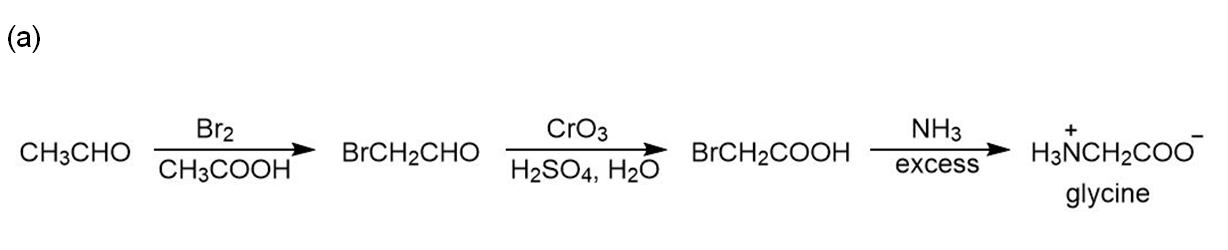
Chapter 29: Q42. (page 1193)
Devise a synthesis of each amino acid from acetaldehyde:
(a) Glycine;
(b) Alanine.
Short Answer
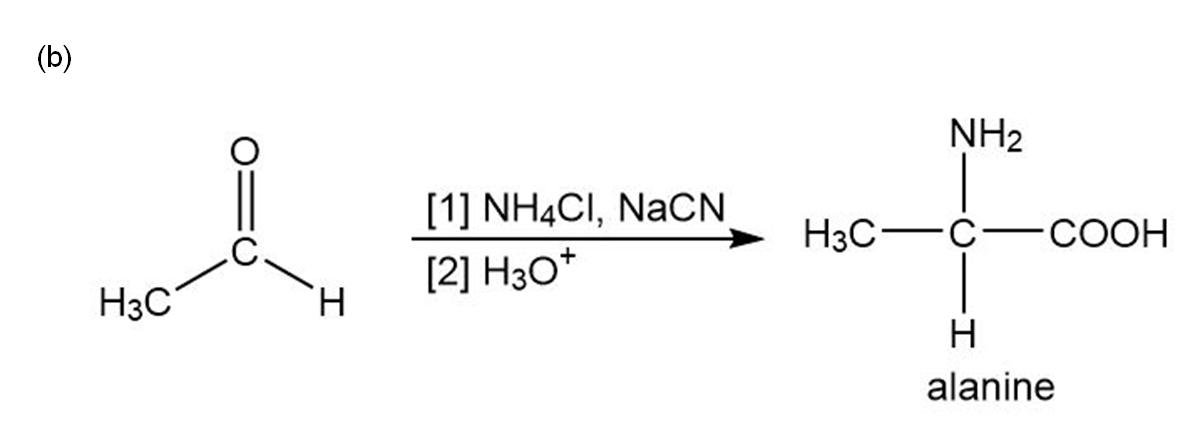
Learning Materials
Features
Discover
Chapter 29: Q42. (page 1193)
Devise a synthesis of each amino acid from acetaldehyde:
(a) Glycine;
(b) Alanine.

All the tools & learning materials you need for study success - in one app.
Get started for free
What - halo carbonyl compoundis needed to synthesize each amino acid?
(a) Glycine
(b) Isoleucine
(c) Phenylalanine
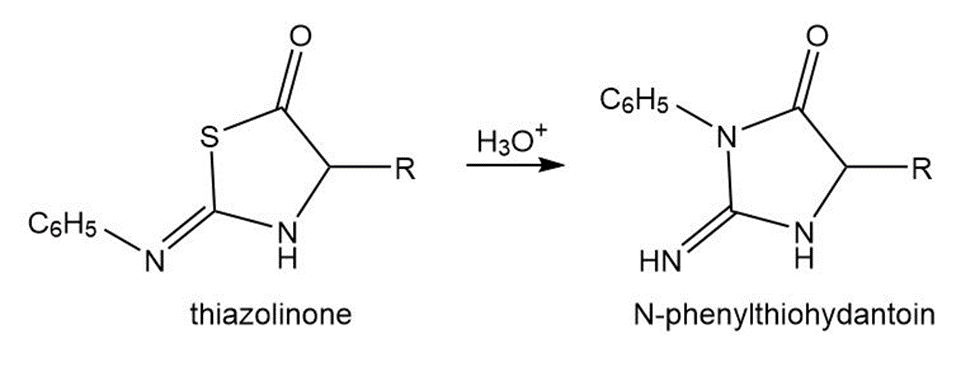
As shown in Mechanism 29.2, the final steps in the Edman degradation result in rearrangement of a thiazolinone to an N-phenylthiohydantoin. Draw a stepwise mechanism for this acid-catalyzed reaction.
Outline the steps needed to synthesize the tetrapeptideAla–Leu–Ile–Gly using the Merrifield technique.
Write out a scheme for the resolution of the two enantiomers of the antiplatelet drug clopidogrel with 10-camphorsulfonic acid.
 Clopidogrel 10-camphorsulfonic acid
Clopidogrel 10-camphorsulfonic acid
Draw the organic products formed in the following reaction.
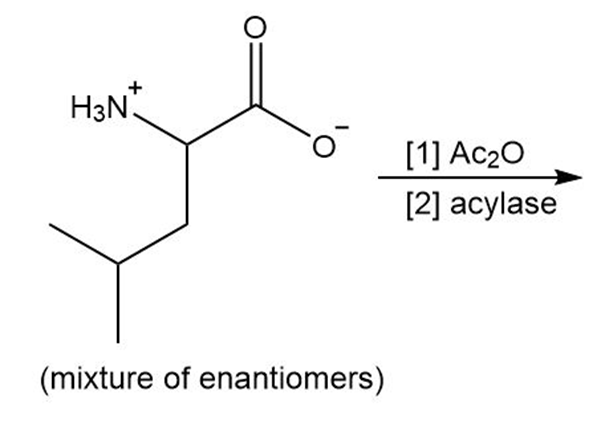
What do you think about this solution?
We value your feedback to improve our textbook solutions.