Chapter 29: Q41P (page 1193)
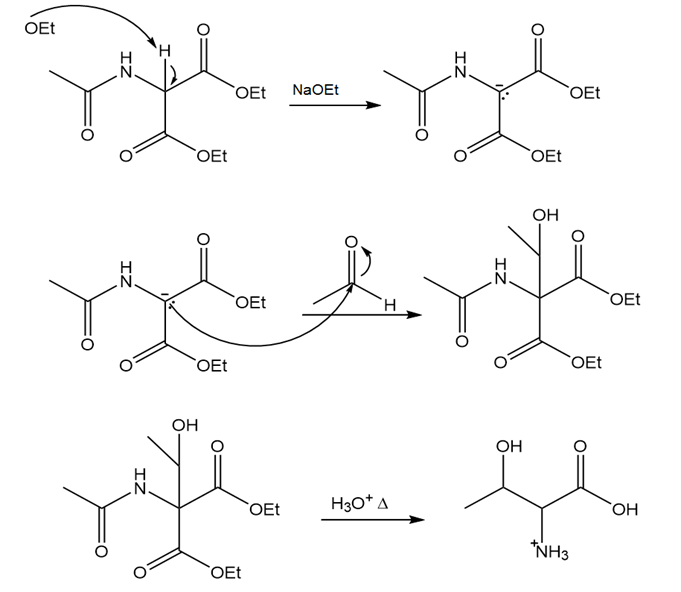
Devise a synthesis of threonine from diethyl acetamidomalonate.
Short Answer
Learning Materials
Features
Discover
Chapter 29: Q41P (page 1193)
Devise a synthesis of threonine from diethyl acetamidomalonate.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Propose a structure for a compound of molecular formula \({{\bf{C}}_{\bf{7}}}{{\bf{H}}_{{\bf{14}}}}{{\bf{O}}_{\bf{2}}}\) with an IR absorption at \({\bf{1740}}\;{\bf{c}}{{\bf{m}}^{{\bf{ - 1}}}}\) and the following \({}^{\bf{1}}{\bf{H}}\) NMR data:
\(\begin{array}{*{20}{c}}{{\bf{Absorption}}}&{{\bf{ppm}}}&{{\bf{Relative}}\;{\bf{area}}}\\\hline{{\bf{singlet}}}&{{\bf{1}}{\bf{.2}}}&{\bf{9}}\\{{\bf{triplet}}}&{{\bf{1}}{\bf{.3}}}&{\bf{3}}\\{{\bf{quartet}}}&{{\bf{4}}{\bf{.1}}}&{\bf{2}}\end{array}\)
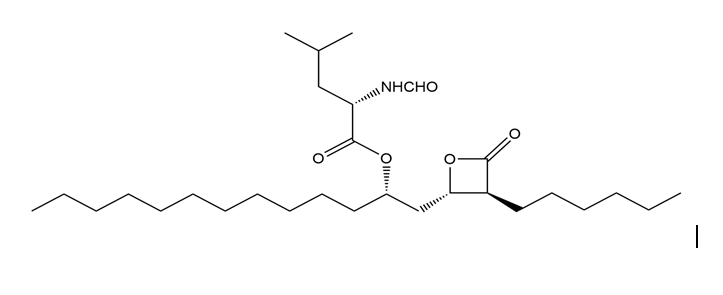
The anti-obesity drug orlistat works by irreversibly inhibiting pancreatic lipase, an enzyme responsible for the hydrolysis of triacylglycerols in the intestines, so they are excreted without metabolism, inhibition occurs by reaction of orlistat with a serine residue of the enzyme, forming a covalently bound, inactive enzyme product. Draw the structure of the product formed during inhibition.
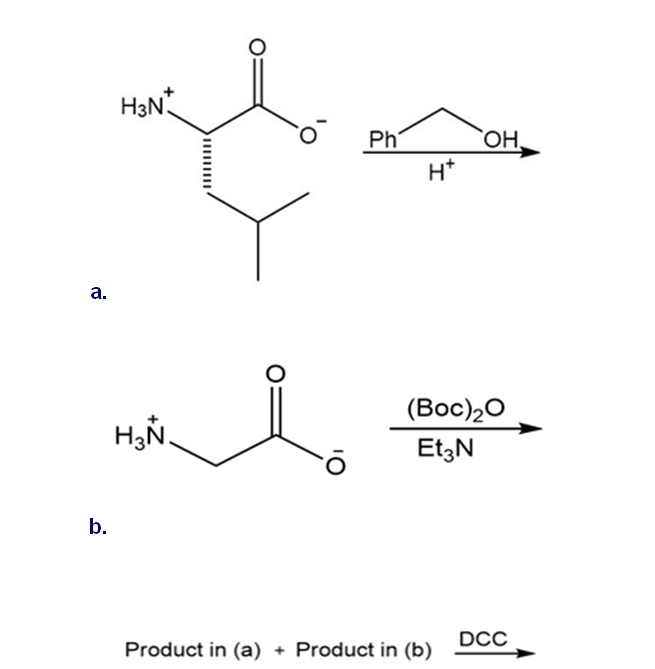
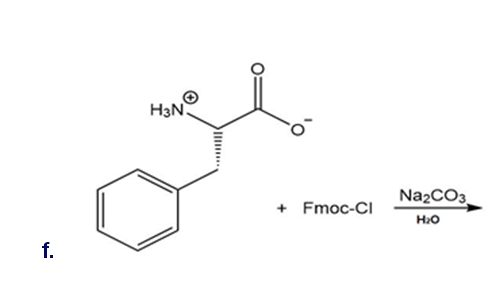
Draw the organic products formed in each reaction.
Question: For the tetra peptide Asp–Arg–Val–Tyr:
a. Name the peptide using one-letter abbreviations.
b. Draw the structure.
c. Label all amide bonds.
d. Label the N-terminal and C-terminal amino acids
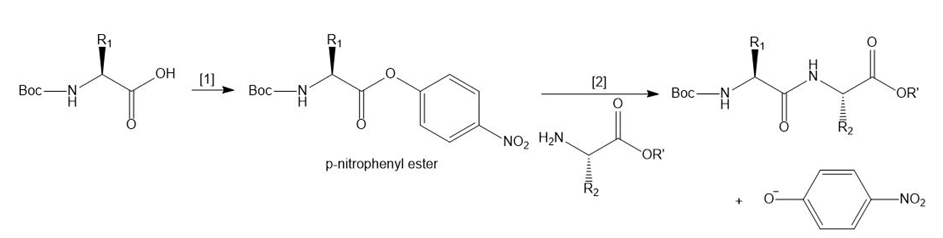
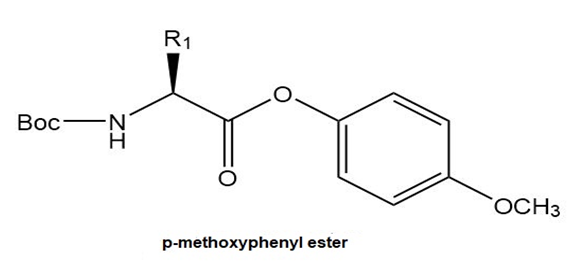
Another method to form a peptide bond involves a two-step process:
[1] Conversion of a Boc-protected amino acid to a -nitrophenyl ester.
[2] Reaction of the -nitrophenyl ester with an amino acid ester
What do you think about this solution?
We value your feedback to improve our textbook solutions.