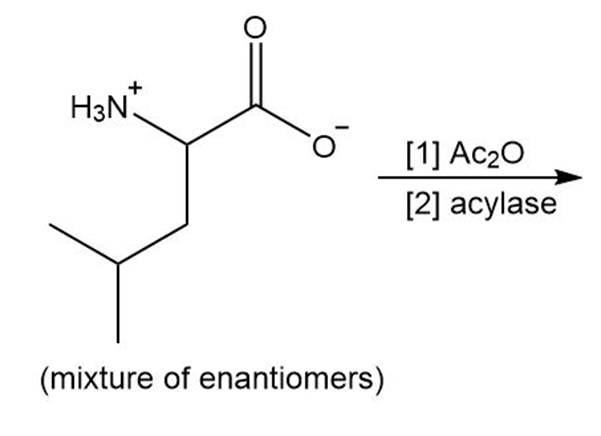
Chapter 29: Q12. (page 1163)
Draw the organic products formed in the following reaction.
Learning Materials
Features
Discover
Chapter 29: Q12. (page 1163)
Draw the organic products formed in the following reaction.

All the tools & learning materials you need for study success - in one app.
Get started for free
Which of the following amines can be used to resolve a racemic mixture of amino acids?
(a)
(b)
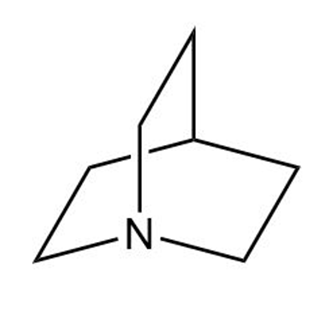
(c)
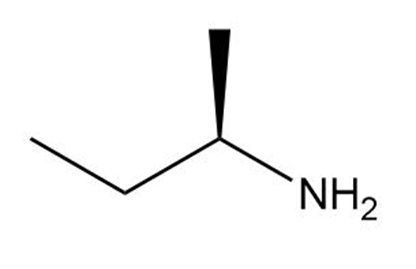
(d)
Question: Propose a structure for a compound of molecular formula \({{\bf{C}}_{\bf{7}}}{{\bf{H}}_{{\bf{14}}}}{{\bf{O}}_{\bf{2}}}\) with an IR absorption at \({\bf{1740}}\;{\bf{c}}{{\bf{m}}^{{\bf{ - 1}}}}\) and the following \({}^{\bf{1}}{\bf{H}}\) NMR data:
\(\begin{array}{*{20}{c}}{{\bf{Absorption}}}&{{\bf{ppm}}}&{{\bf{Relative}}\;{\bf{area}}}\\\hline{{\bf{singlet}}}&{{\bf{1}}{\bf{.2}}}&{\bf{9}}\\{{\bf{triplet}}}&{{\bf{1}}{\bf{.3}}}&{\bf{3}}\\{{\bf{quartet}}}&{{\bf{4}}{\bf{.1}}}&{\bf{2}}\end{array}\)
What is the structure of each amino acid at its isoelectric point: (a) alanine (b) methionine; (c)aspartic acid; (d) lysine
Question: Into how many peaks will each proton shown in green be split?
a.
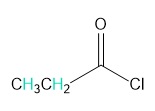
b.
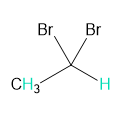
c.
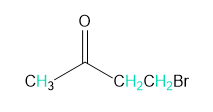
d.
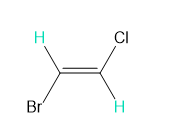
e.
 Image Caption
Image Caption
f.
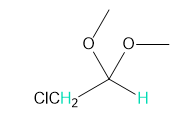
Draw the structure for each peptide:
(a) Phe–Ala;
(b) Gly–Gln;
(c) Lys–Gly;
(d) R-H
What do you think about this solution?
We value your feedback to improve our textbook solutions.