Chapter 25: 48 (page 1043)
What amide(s) can be used to prepare each amine by reduction?
(a)
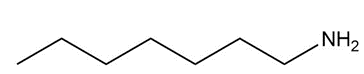
(b)
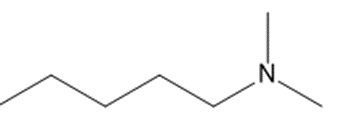
(c)
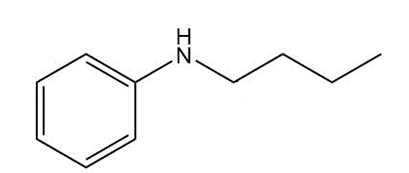
Short Answer
(a)
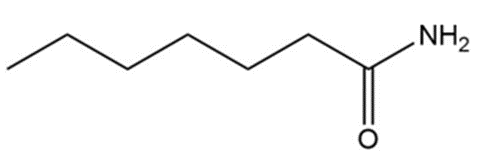
(b)
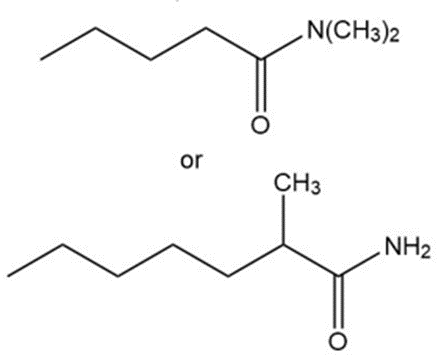
(c)
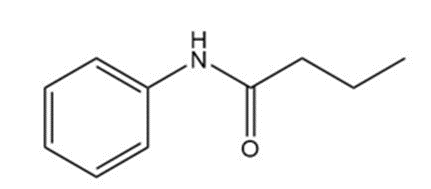
Learning Materials
Features
Discover
Chapter 25: 48 (page 1043)
What amide(s) can be used to prepare each amine by reduction?
(a)

(b)

(c)

(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
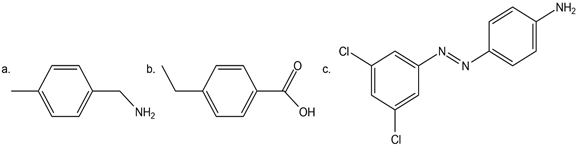
Question: Synthesize each compound from benzene. Use a diazonium salt as one of the synthetic intermediates.
Question: What is the structure of an unknown compound with molecular formula C6H15N that gives the following 1H NMR absorptions: 0.9 (singlet, 1 H), 1.10 (triplet, 3 H), 1.15 (singlet, 9 H), and 2.6 (quartet, 2 H) ppm?
Question: Write out steps to show how each of the following pairs of compounds can be separated by an extraction procedure.
a.
 and
and 
b.
 and
and 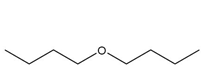
Question: Draw the product formed in each reaction.
a.
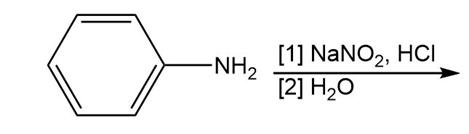
b.
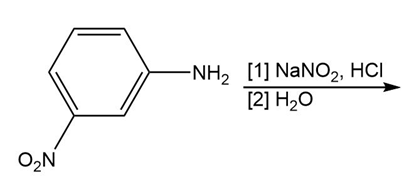
c.
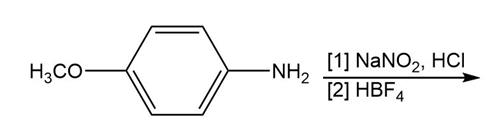
d.
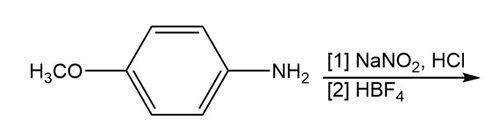
Question: Draw the product formed when C6H5N2+Cl– reacts with each compound.
a.
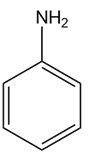
b.
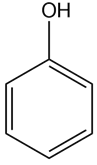
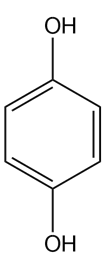
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.