Chapter 25: 47 (page 1042)
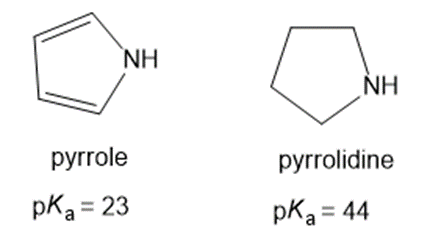
Why is pyrrole more acidic than pyrrolidine?
Short Answer
Pyrrole is more acidic than pyrrolidine because its conjugate base is more stable than pyrrolidine.
Learning Materials
Features
Discover
Chapter 25: 47 (page 1042)
Why is pyrrole more acidic than pyrrolidine?

Pyrrole is more acidic than pyrrolidine because its conjugate base is more stable than pyrrolidine.
All the tools & learning materials you need for study success - in one app.
Get started for free
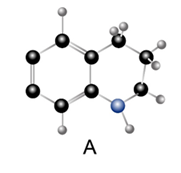
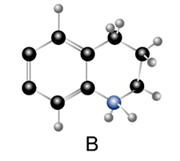
Question: Which compound is the stronger base?
Question: Draw the structure that corresponds to each name.
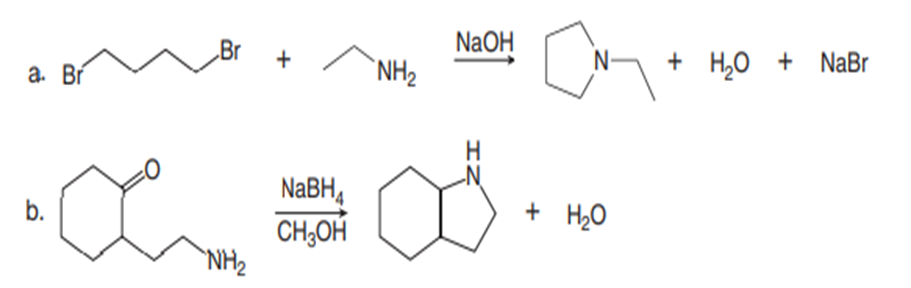
Draw a stepwise mechanism for each reaction.
Question: Draw a structure corresponding to each name.
a. 2,4-dimethylhexan-3-amine
b. N-methylpentan-1-amine
c. N-isopropyl-p-nitroaniline
d. N-methylpiperidine
e. N,N-dimethylethanaminef. 2-aminocyclohexanoneg. N-methylanilineh. m-ethylaniline
Question: Draw the product of each reaction.
a.
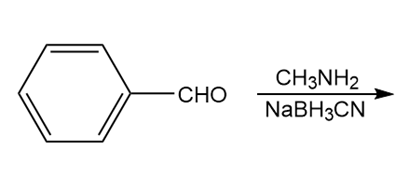
b.
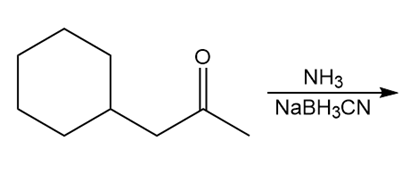
c.
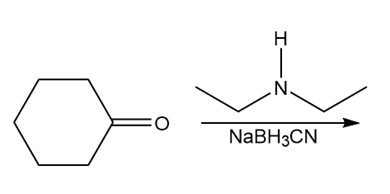
d.
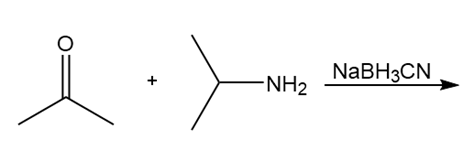
What do you think about this solution?
We value your feedback to improve our textbook solutions.