Chapter 11: Q39. (page 451)
Question: Draw the structure of compounds A–E in the following reaction scheme:
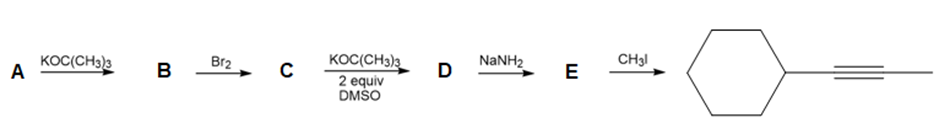
Short Answer
Answer:
Learning Materials
Features
Discover
Chapter 11: Q39. (page 451)
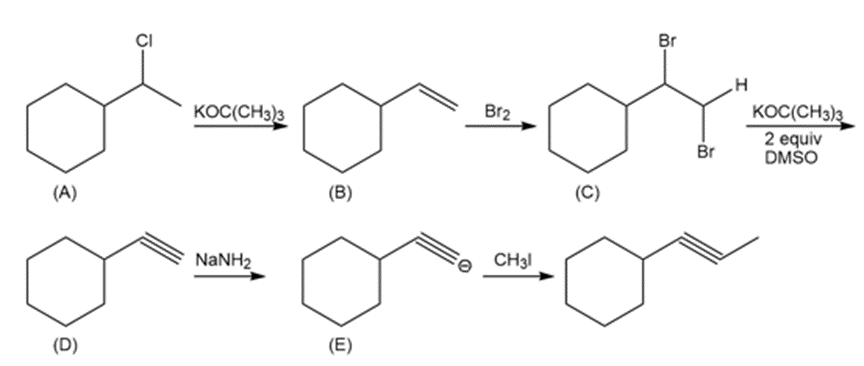
Question: Draw the structure of compounds A–E in the following reaction scheme:

Answer:

All the tools & learning materials you need for study success - in one app.
Get started for free
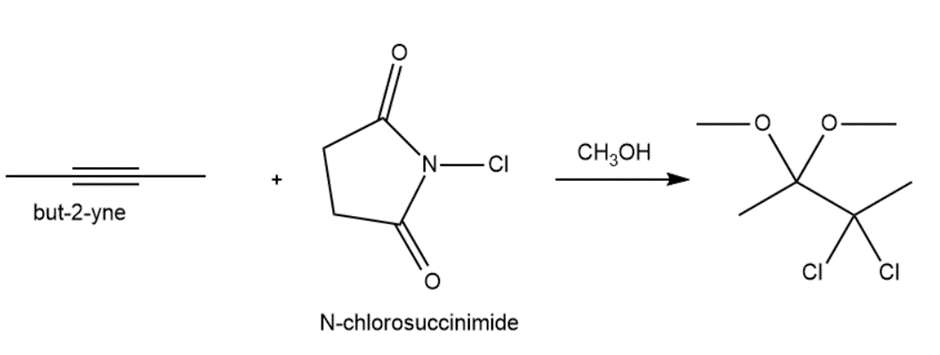
Question: N-Chlorosuccinimide (NCS) serves as a source of Cl+in electrophilic addition reactions to alkenes and alkynes. Keeping this in mind, draw a stepwise mechanism for the following addition to buy-2-yne.
Question: Draw the enol tautomer of (a) and the keto tautomer of (b).
a.
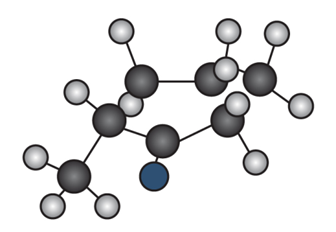
b. 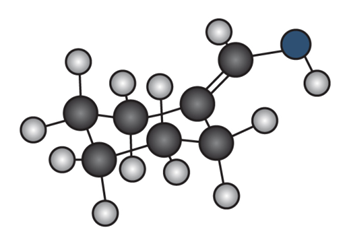
Which bases can deprotonate acetylene? The pKa values of the conjugate acids are given in parentheses.
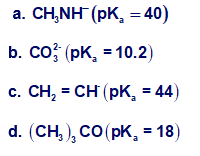
Question: What acetylide anion and alkyl halide can be used to prepare each alkyne? Indicate all possibilities when more than one route will work.
a.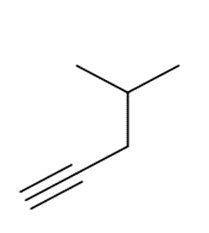
b.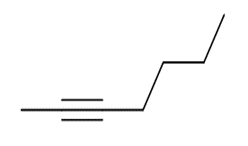
c.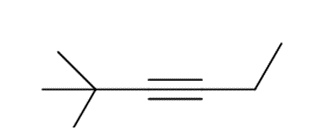
Question. Give the structure corresponding to each of the following names.
a. trans-2-ethynylcyclopentanol
b. 4-tert-butyldec-5-yne
c. 3,3,5-trimethylcyclononyne
What do you think about this solution?
We value your feedback to improve our textbook solutions.