Chapter 11: Q38. (page 450)
Question: Draw the organic products formed in each reaction.
a. 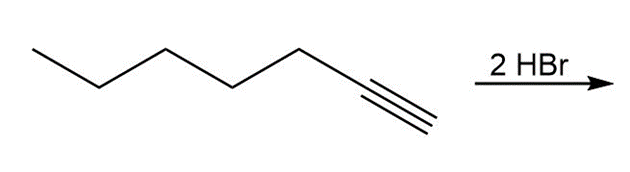
b. 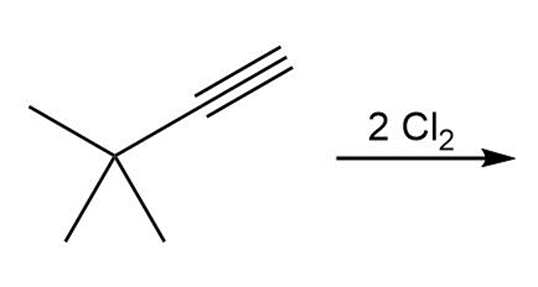
c. 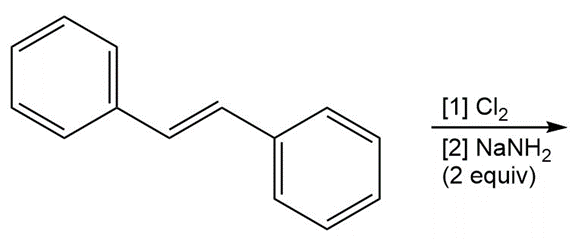
d. 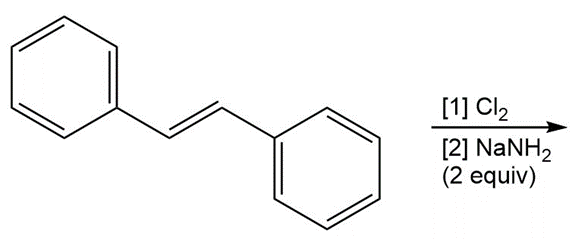
e. 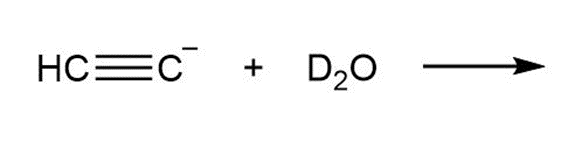
f.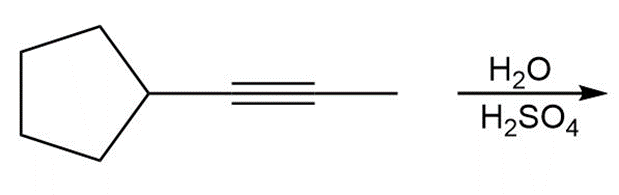
g.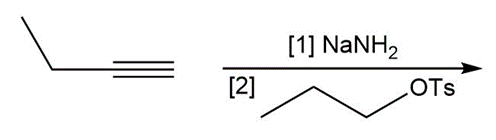
h. 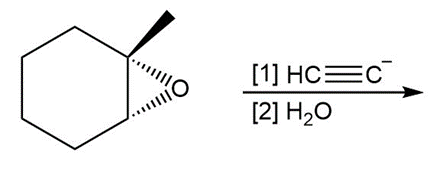
i. 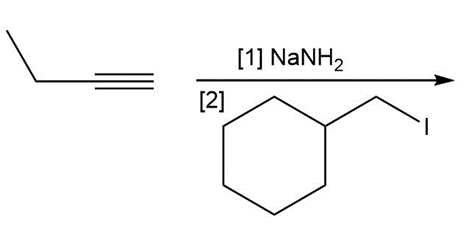
j. 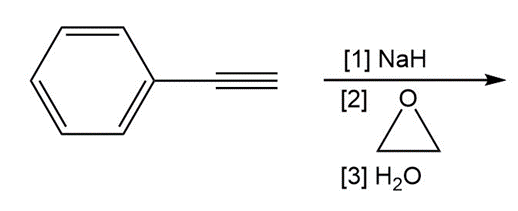
Short Answer
a. 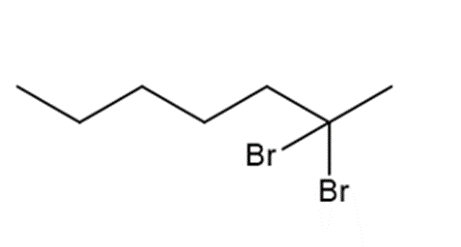
b. 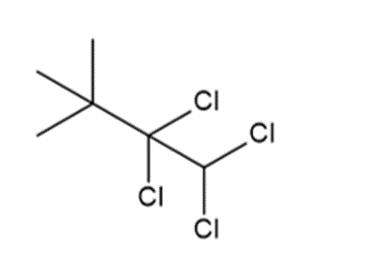
c 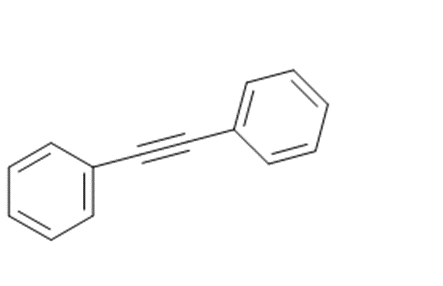
d. 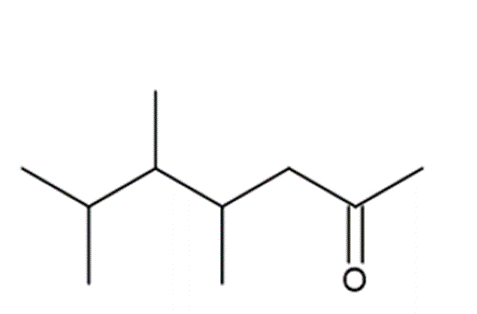
e. 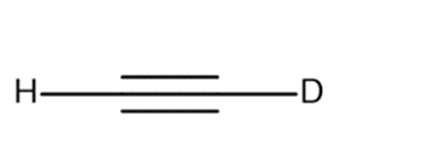
f. 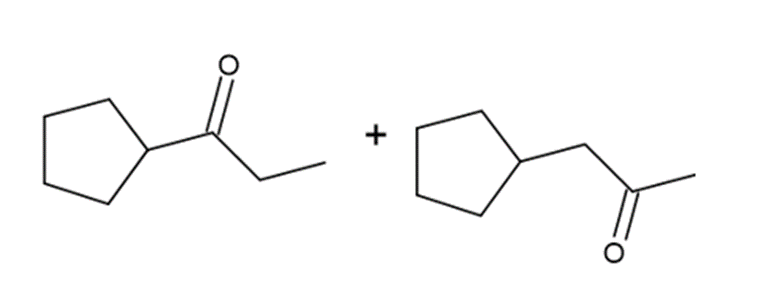
g. 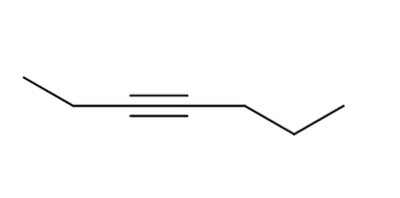
h. 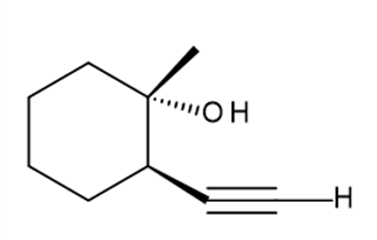
i.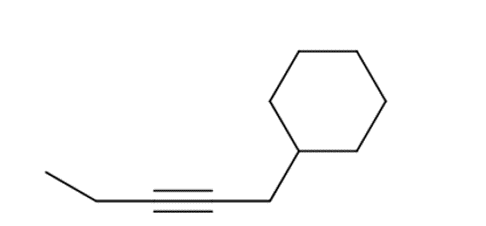
j.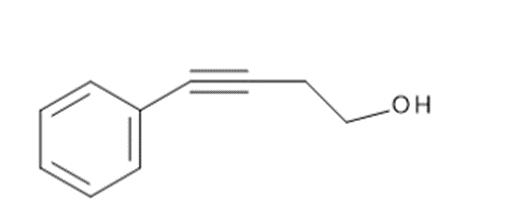
Learning Materials
Features
Discover
Chapter 11: Q38. (page 450)
Question: Draw the organic products formed in each reaction.
a. 
b. 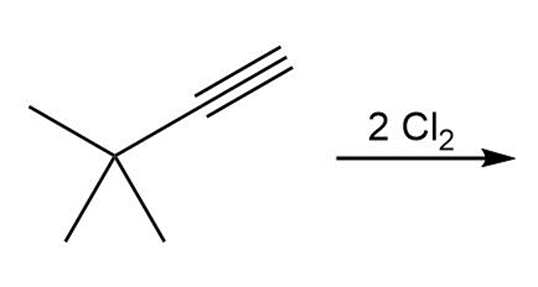
c. 
d. 
e. 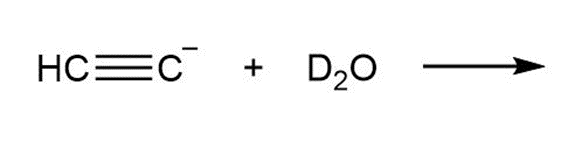
f.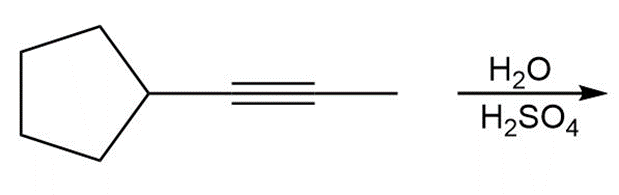
g.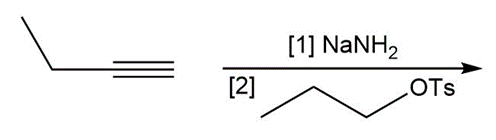
h. 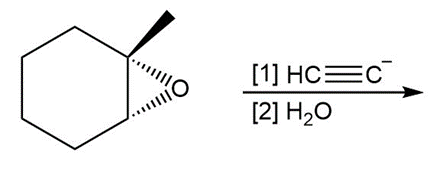
i. 
j. 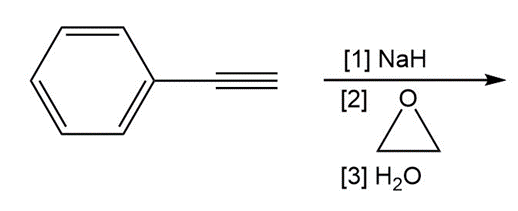
a. 
b. 
c 
d. 
e. 
f. 
g. 
h. 
i.
j.
All the tools & learning materials you need for study success - in one app.
Get started for free
Convert each compound to hex-1-yne,
a.b.
c.
Question: From what you have learned about enols and the hydration of alkynes, predict what product is formed by the acid-catalyzed hydration of . Draw a stepwise mechanism that illustrates how it is formed.
Question: Explain why 2,2,5,5-tetramethylhex-3-yne can’t be made using acetylide anions
Question: Devise a synthesis of the ketone hexan-3-one, CH3CH2COCH2CH2CH3 , from CH3CH2Br as the only organic starting material; that is, all the carbon atoms in hexan-3-one must come from CH3CH2Br . You may use only other needed reagents.
Question: Explain the apparent paradox. Although the addition of one equivalent of HX to an alkyne is more exothermic than the addition of HX to an alkene, an alkene reacts faster with HX.
What do you think about this solution?
We value your feedback to improve our textbook solutions.