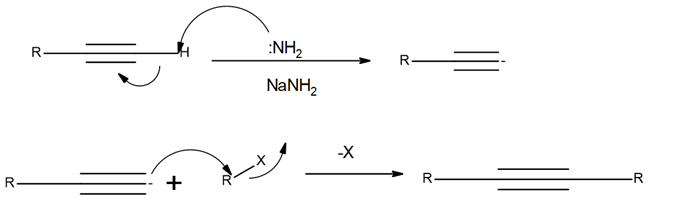
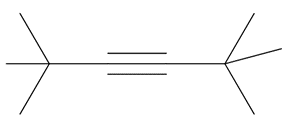
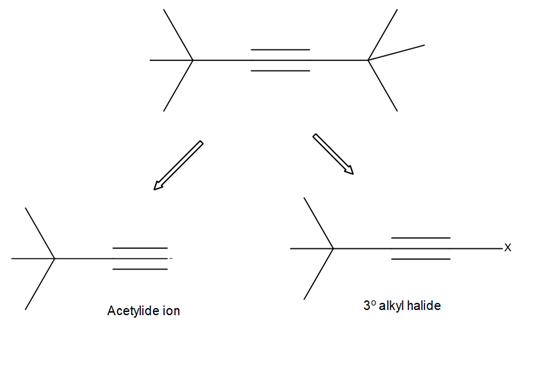
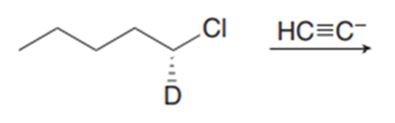
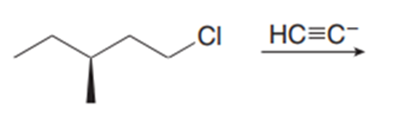
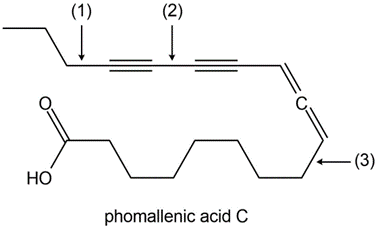
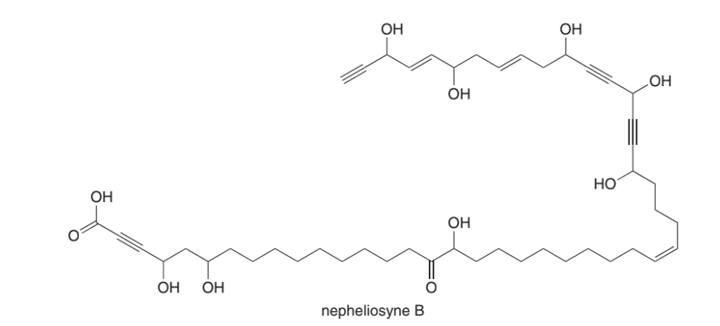
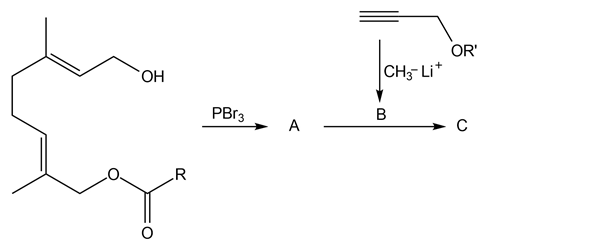
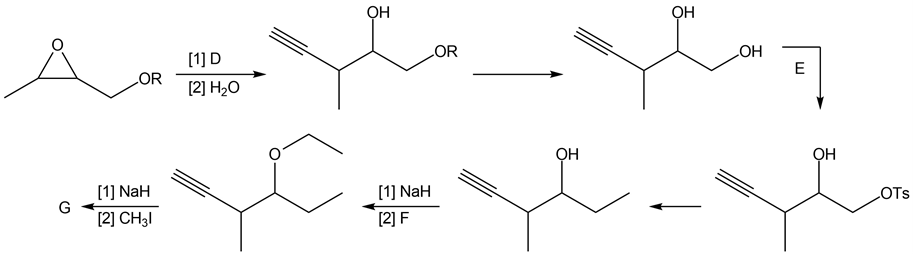
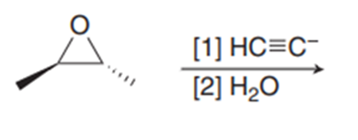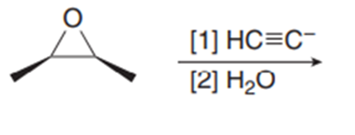Chapter 11: Q18. (page 444)
Question: Explain why 2,2,5,5-tetramethylhex-3-yne can’t be made using acetylide anions
Short Answer
Answer
- The carbon centre is too crowded for the acetylide anion to attack.
- Elimination would be preferred over substitution