Chapter 11: 5P (page 426)
Convert each compound to hex-1-yne,
a.b.
c.
Short Answer
a.
b.
c.
.
Learning Materials
Features
Discover
Chapter 11: 5P (page 426)
Convert each compound to hex-1-yne,
a.b.
c.
a.
b.
c.
.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What reagents are needed to convertto each compound?
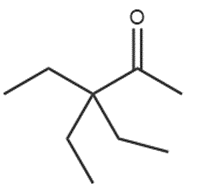
a.
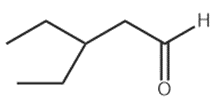
b.
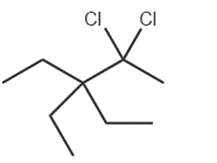
c.
d.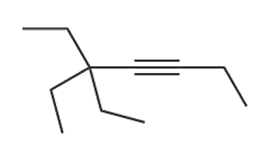
Question: Devise a synthesis of from two-carbon starting materials.
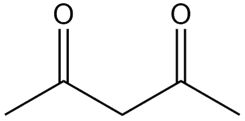
Question: How is each compound related to A? Choose from tautomers, constitutional isomers but not tautomers, or neither
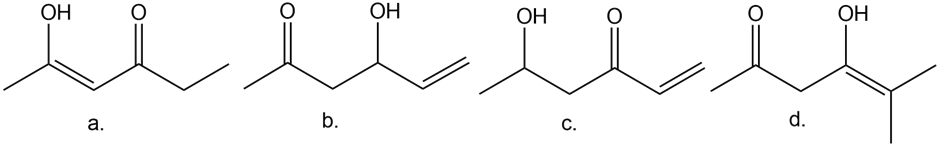
A.
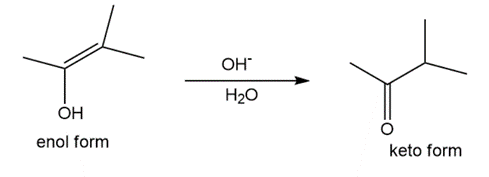
Question: Tautomerization in base resembles tautomerization in acid, but deprotonation precedes protonation in the two-step mechanism. (a) Draw a stepwise mechanism for the following tautomerization. (b) Then, draw a stepwise mechanism for the reverse reaction, the conversion of the keto form to the enol.
Question:
(a) Draw two different enol tautomers of 2-methylcyclohexanone.
(b) Draw two constitutional isomers that are not tautomers, but contain a and an O-H group.
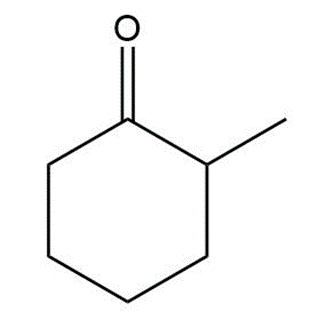
2-methylcyclohexanone
What do you think about this solution?
We value your feedback to improve our textbook solutions.