Chapter 11: 6P (page 426)
Which bases can deprotonate acetylene? The pKa values of the conjugate acids are given in parentheses.
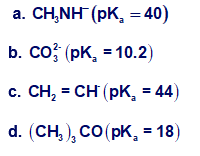
Short Answer
Bases that can deprotonate acetylene are
Learning Materials
Features
Discover
Chapter 11: 6P (page 426)
Which bases can deprotonate acetylene? The pKa values of the conjugate acids are given in parentheses.
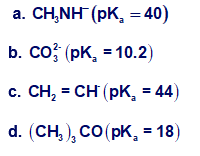
Bases that can deprotonate acetylene are
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Explain the apparent paradox. Although the addition of one equivalent of HX to an alkyne is more exothermic than the addition of HX to an alkene, an alkene reacts faster with HX.
Question: What reagents are needed to convertto each compound?
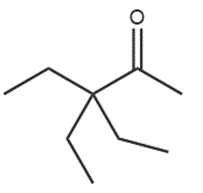
a.
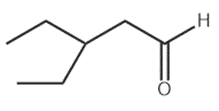
b.
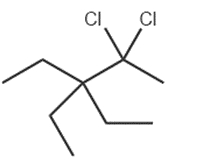
c.
d.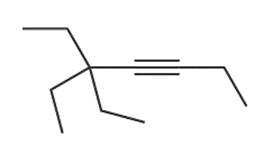
Question: Draw the products formed when the following alkynes are treated with each set of reagents: [1] .
a.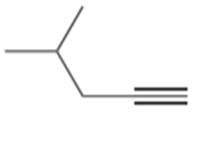
b.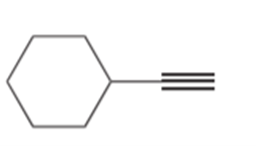
Question: Draw the products of each reaction.
a.
b. 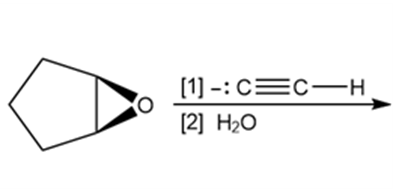
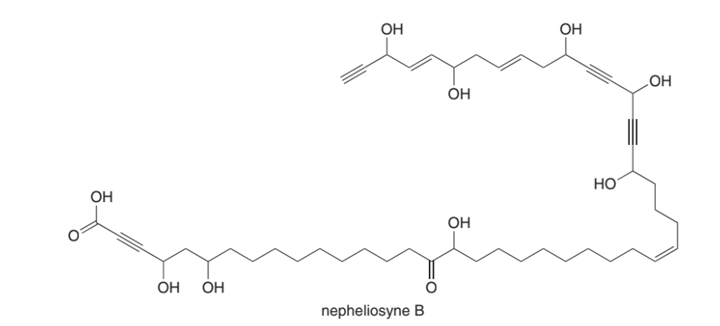
Nepheliosyne B is a novel acetylenic fatty acid isolated from a New Caledonian marine sponge.
(a) Label the most acidic H atom.
(b) Which carbon-carbon σ bond is shortest?
(c) How many degrees of unsaturation does nepheliosyne B contain?
(d) How many bonds are formed from ?
(e) Label each triple bond as internal or terminal.
What do you think about this solution?
We value your feedback to improve our textbook solutions.