Chapter 11: 3P (page 426)
Question. Give the structure corresponding to each of the following names.
a. trans-2-ethynylcyclopentanol
b. 4-tert-butyldec-5-yne
c. 3,3,5-trimethylcyclononyne
Short Answer
a.
b.
c.
Learning Materials
Features
Discover
Chapter 11: 3P (page 426)
Question. Give the structure corresponding to each of the following names.
a. trans-2-ethynylcyclopentanol
b. 4-tert-butyldec-5-yne
c. 3,3,5-trimethylcyclononyne
a.
b.
c.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Identify the lettered compounds in the following reaction schemes. Each reaction sequence was used in the synthesis of a natural product.
a. 
b.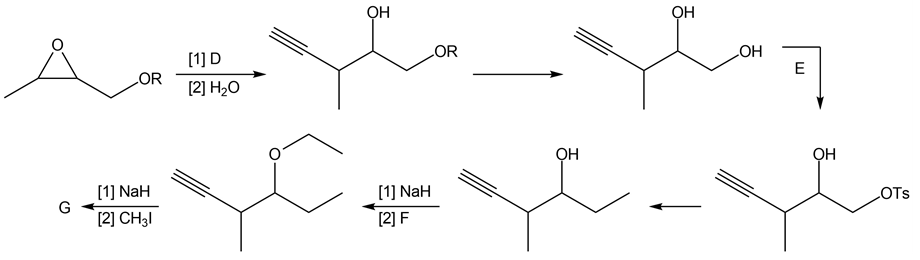
Question: When alkyne a is treated with followed by , a product having the molecular formula is formed, but it is not compound B. What is the structure of the product and why is it formed?

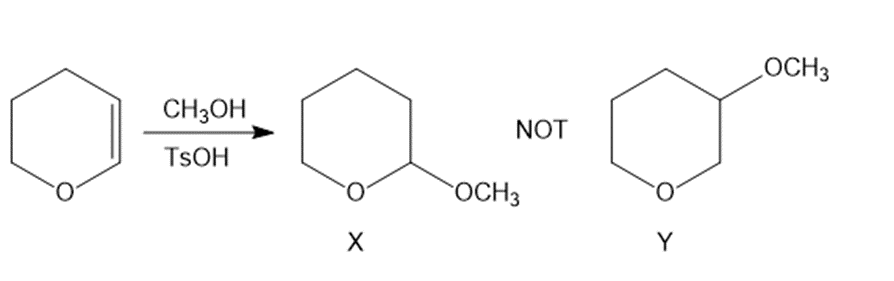
Question: Why is compound X formed in the following reaction instead of its constitutional isomer Y?
Explain the following result. Although alkenes are generally more reactive than alkynes towards electrophiles, the reaction of with but-2-yne can be stopped after one equivalent of has been added.
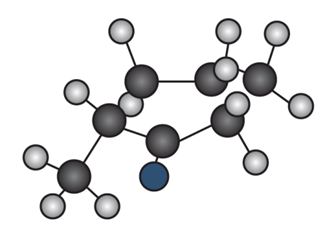
Question: Draw the enol tautomer of (a) and the keto tautomer of (b).
a.
b. 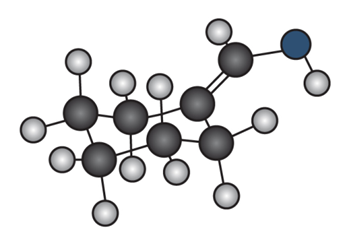
What do you think about this solution?
We value your feedback to improve our textbook solutions.