Chapter 10: Q.22. (page 405)
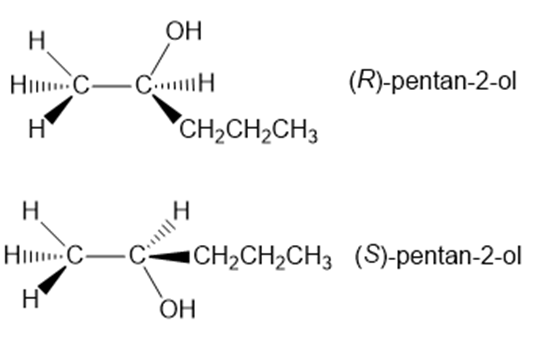
Question: What stereoisomers are formed when pent-1-ene is treated with and ?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 10: Q.22. (page 405)
Question: What stereoisomers are formed when pent-1-ene is treated with and ?
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
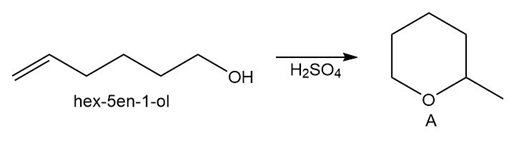
Question: Draw a stepwise mechanism for the conversion of hex-5-en-1-ol to the cyclic ether A.
Question: Give the IUPAC name for each polyfunctional compound.
Question: What three alkenes (excluding stereoisomers) can be used to prepare 3-chloro-3-methylhexane by addition of HCl?
Question: a. What product(s) are formed when the E isomer of C6H5CH=CHC6H5is treated with Br2 , followed by one equivalent of KOH? Label the resulting alkene(s) as E or Z. b. What product(s) are formed when the Z isomer of C6H5CH=CHC6H5 is subjected to the same reaction sequence? c. How are the compounds in parts (a) and (b) related to each other?
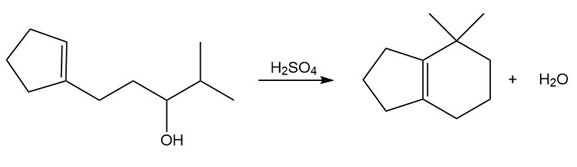
Question: Draw a stepwise mechanism for the following reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.