Chapter 10: PROBLEM 10.70 (page 425)
Question: Draw a stepwise mechanism for the following reaction.
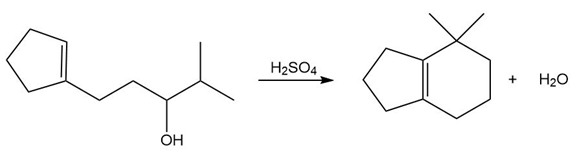
Short Answer
Answer
Learning Materials
Features
Discover
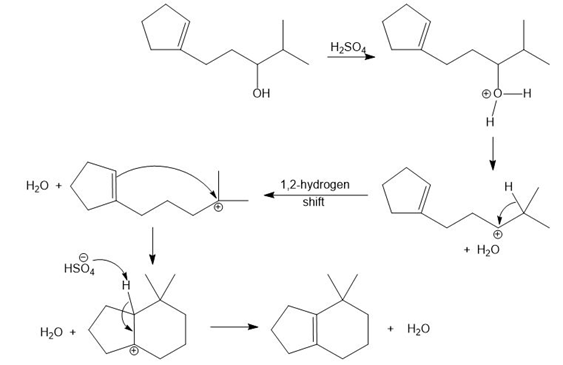
Chapter 10: PROBLEM 10.70 (page 425)
Question: Draw a stepwise mechanism for the following reaction.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How many rings and π bonds does a compound with molecular formula C10H14 possess? List all possibilities.
Question: Label each C-C double bond as E or Z. Kavain is a naturally occurring relaxant isolated from kava root.
Question: Draw the products, including stereochemistry, of each reaction.
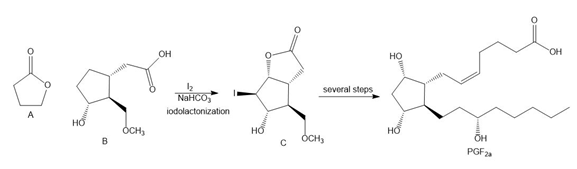
Question: Lactones, cyclic esters such as compound A, are prepared by halolactonization, an addition reaction to an alkene. For example, iodolactonization of B forms lactone C, a key intermediate in the synthesis of prostaglandin (Section 4.15). Draw a stepwise mechanism for this addition reaction.
Question: What alkene can be used to prepare each alkyl halide or dihalide as the exclusive or major product of an addition reaction?
a.
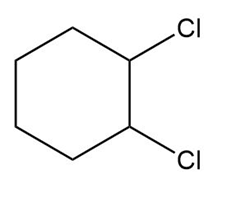
b.
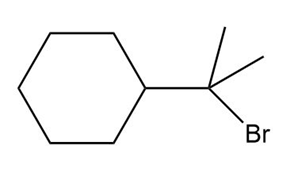
c.
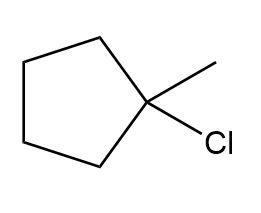
d.
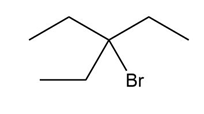
What do you think about this solution?
We value your feedback to improve our textbook solutions.