Chapter 10: Q.12. (page 396)
Question: Draw the products of each elimination reaction.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 10: Q.12. (page 396)
Question: Draw the products of each elimination reaction.
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How many rings and π bonds does a compound with molecular formula C10H14 possess? List all possibilities.
Question: Draw the six alkenes of the molecular formula C5H10. Label one pair of diastereomers.
Question: Give the structure corresponding to each name.
a. (E)-4-ethylhept-3-ene
b. 3,3-dimethylcyclopentene
c. 4-vinylcyclopentene
d. (Z)-3-isopropylhept-2-ene
e. cis-3,4-dimethylcyclopentene
f. 1-isopropyl-4-propylcyclohexene
g. 3,4-dimethylcyclohex-2-enol
h. 3,5-diethylhex-5-en-3-ol
Question: Treatment of 3-methylcyclohexene with HCl yields two products, 1-chloro-3-methylcyclohexane and 1-chloro-1-methylcyclohexane. Draw a mechanism to explain this result.
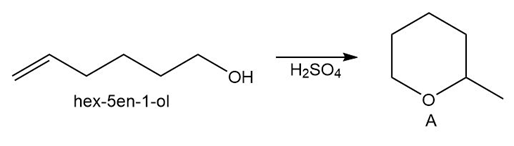
Question: Draw a stepwise mechanism for the conversion of hex-5-en-1-ol to the cyclic ether A.
What do you think about this solution?
We value your feedback to improve our textbook solutions.