Chapter 10: PROBLEM 10.42 (page 421)
Question: Name each of the following epoxides as an alkene oxide.
a.
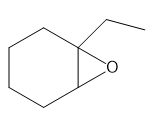
b.
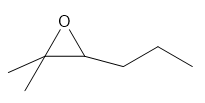
c.
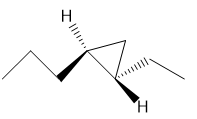
d.

Short Answer
Answer
a. 1-ethylcyclohexene oxide
b. 2-methyl-2-hexene oxide
c. Trans-3-heptene oxide
d. 4-tert-butylcyclopentene oxide
Learning Materials
Features
Discover
Chapter 10: PROBLEM 10.42 (page 421)
Question: Name each of the following epoxides as an alkene oxide.
a.

b.

c.

d.

Answer
a. 1-ethylcyclohexene oxide
b. 2-methyl-2-hexene oxide
c. Trans-3-heptene oxide
d. 4-tert-butylcyclopentene oxide
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the constitutional isomer formed in each reaction.
a.
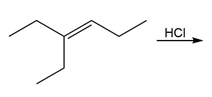
b.
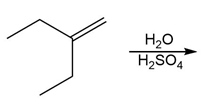
c.
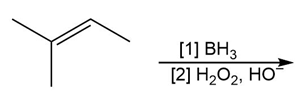
d.
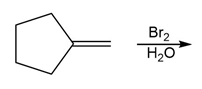
e.
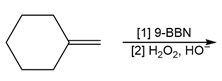
f.
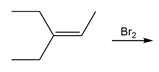
Question: What stereoisomers are formed when pent-1-ene is treated with and ?
Question: (a) Draw the structure of (1E,4R)-1,4-dimethylcyclodecene. (b) Draw the enantiomer and name it, including its E,Z and R,S prefixes. (c) Draw two diastereomers and name them, including the E,Z and R,S prefixes.
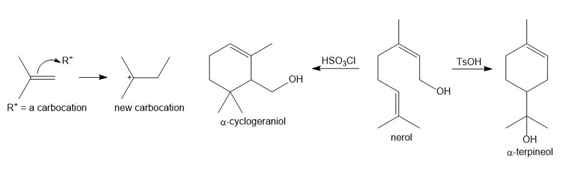
Question: Like other electrophiles, carbocations add to alkenes to form new carbocations, which can then undergo substitution or elimination reactions depending on the reaction conditions. With this in mind, consider the following reactions of nerol, a natural product isolated from lemon grass and other plant sources. Treatment of nerol with TsOH forms α-terpineol as the major product, whereas treatment of nerol with chlorosulfonic acid, HSO3Cl , forms a constitutional isomer, α-cyclogeraniol. Write stepwise mechanisms for both processes. Each mechanism involves the addition of an electrophile—a carbocation— to a double bond.
Question: Draw the products of each elimination reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.