Chapter 10: Alkenes
PROBLEM 10.55
Question: Using cis- and trans-hex-3-ene, demonstrate that the addition of HCl is not a stereospecific reaction. Draw the structure of the stereoisomers formed from each alkene.
PROBLEM 10.56
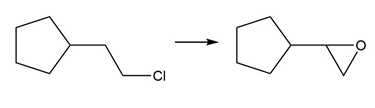
Question: Draw a stepwise mechanism for the following reaction.
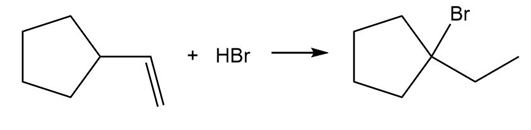
PROBLEM 10.57
Question: Draw a stepwise mechanism for the following reaction, which results in ring expansion of a six-membered ring to a seven-membered ring.
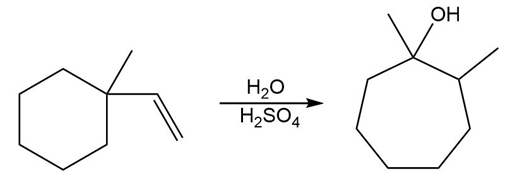
PROBLEM 10.58
Question: Draw a stepwise mechanism for the conversion of hex-5-en-1-ol to the cyclic ether A.
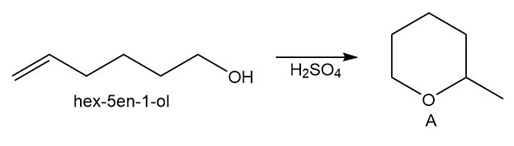
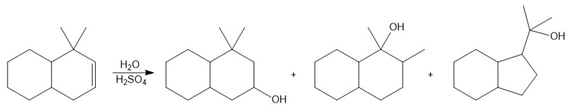
PROBLEM 10.59
Question: Draw a stepwise mechanism that shows how all three alcohols are formed from the bicyclic alkene.
PROBLEM 10.60
Question: Less stable alkenes can be isomerized to more stable alkenes by treatment with strong acid. For example, 2,3-dimethylbut-1-ene is converted to 2,3-dimethylbut-2-ene when treated with . Draw a stepwise mechanism for this isomerization process.
PROBLEM 10.61
Question: When buta-1,3-diene (CH2=CH-CH=CH2) is treated with HBr, two constitutional isomers are formed CH3CHBr-CH=CH2and Br-CH2CH=CHCH2. Draw a stepwise mechanism that accounts for the formation of both products.
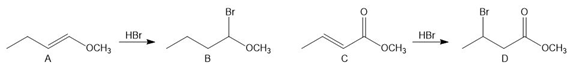
PROBLEM 10.62
Question: Explain why the addition of HBr to alkenes A and C is regioselective, forming addition products B and D, respectively.
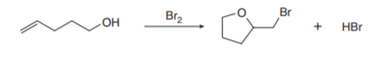
PROBLEM 10.63
Question: Bromoetherification, the addition of the elements of Br and OR to a double bond, is a common method for constructing rings containing oxygen atoms. This reaction has been used in the synthesis of the polyether antibiotic monensin (Problem 21.37). Draw a stepwise mechanism for the following intramolecular bromoetherification reaction.
PROBLEM 10.64
Question: Devise a synthesis of each product from the given starting material. More than one step is required.
a.
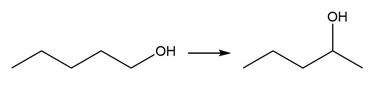
b.
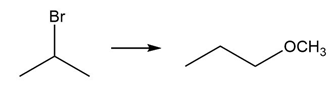
c.
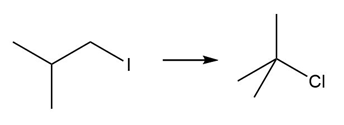
d.
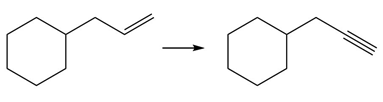
e.
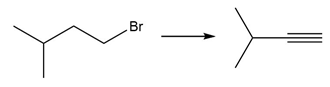
f.