Chapter 10: PROBLEM 10.56 (page 423)
Question: Draw a stepwise mechanism for the following reaction.
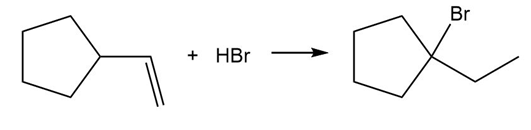
Short Answer
Answer
Learning Materials
Features
Discover
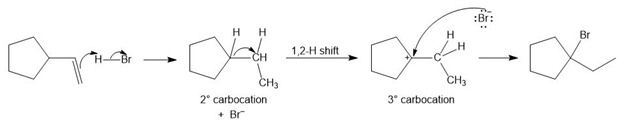
Chapter 10: PROBLEM 10.56 (page 423)
Question: Draw a stepwise mechanism for the following reaction.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Use the Hammond postulate to explain why reacts faster than in electrophilic addition of HX.
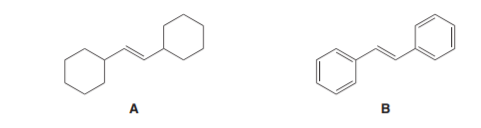
Question: Which alkene reacts faster with HBr? Explain your choice
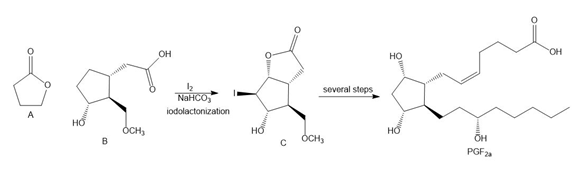
Question: Lactones, cyclic esters such as compound A, are prepared by halolactonization, an addition reaction to an alkene. For example, iodolactonization of B forms lactone C, a key intermediate in the synthesis of prostaglandin (Section 4.15). Draw a stepwise mechanism for this addition reaction.
Question: Draw the products of each elimination reaction.
Question: Draw all stereoisomers formed in each reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.