Chapter 4: Q.60. (page 128)
Question: Classify each pair of compounds as constitutional isomers or identical molecules.
a.
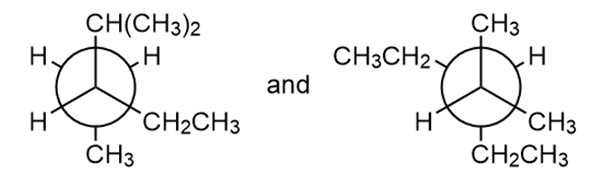
b.
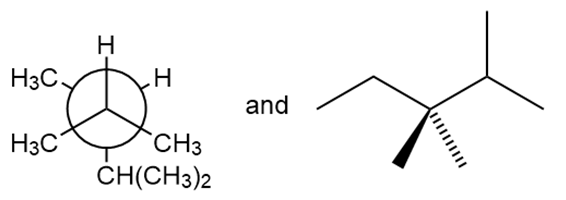
Short Answer
Answer
- The given compounds are identical.
- The given compounds are constitutional isomers.
Learning Materials
Features
Discover
Chapter 4: Q.60. (page 128)
Question: Classify each pair of compounds as constitutional isomers or identical molecules.
a.

b.

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
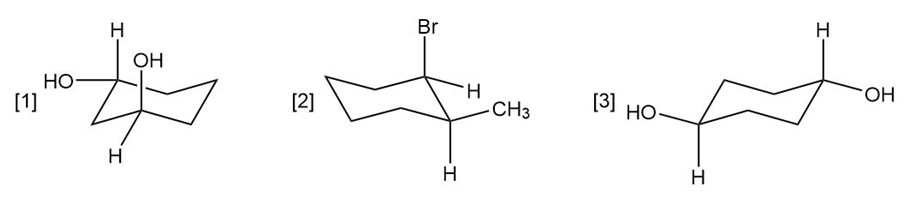
Question: For each compound drawn below:
a. Label each OH, Br, andgroup as axial or equatorial.
b. Classify each conformation as cis or trans.
c. Translate each structure into a representation with a hexagon for the six-membered ring, and wedges and dashed wedges for groups above and below the ring.
d. Draw the second possible chair conformation for each compound.
Question: Draw the five constitutional isomers having molecular formula .
Question: Give the structure and IUPAC name for each of the nine isomers having molecular formula that contains seven carbons in the longest chain and two methyl groups as substituents.
Question:Give IUPAC name of each compound.
a.
b.
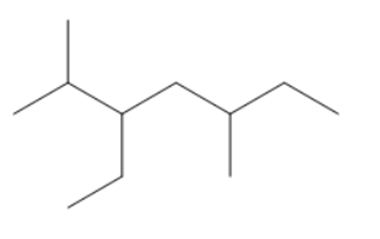
c.
d.
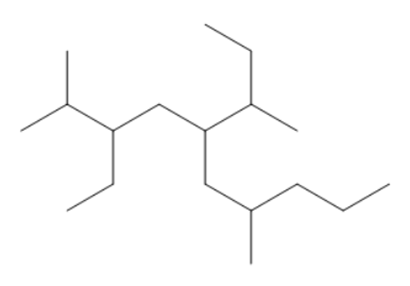
Question: Give the IUPAC name for each compound.
What do you think about this solution?
We value your feedback to improve our textbook solutions.