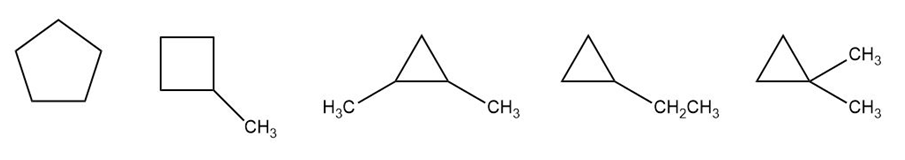
Chapter 4: Q.6. (page 128)
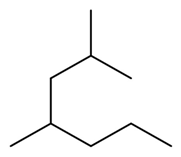
Question: Draw the five constitutional isomers that have molecular formula and contain one ring.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 4: Q.6. (page 128)
Question: Draw the five constitutional isomers that have molecular formula and contain one ring.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Cyclopropane and cyclobutane have similar strain energy despite the fact that the C-C-C bond angles of cyclopropane are much smaller than those of cyclobutane. Suggest an explanation for this observation, considering all sources of strain discussed in Chapter 4.
Question: The torsional energy in propane is 14 kJ/mol (3.4 kcal/mol). Because each H,H eclipsing interaction is worth 4.0 kJ/mol (1.0 kcal/mol) of destabilization, how much is one H, eclipsing interaction worth in destabilization? (See Section 4.10 for an alternate way to arrive at this value.)
Question: Draw both conformations for 1-ethyl-1-methylcyclohexane and decide which conformation (if any) is more stable.
Question: Give IUPAC name of each compound.
a.
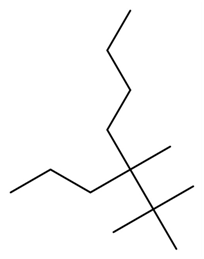
b.
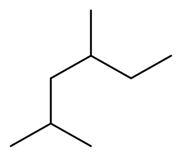
c.
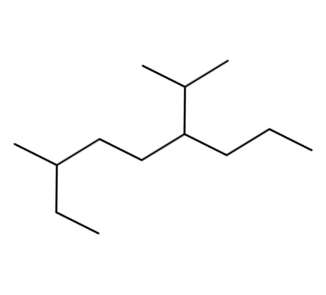
d.
Question: Consider the tricyclic structure B.
(a) Label each substituent on the rings as axial or equatorial.
(b) Draw B using chair conformations for each six-membered ring.
(c) Label the atoms on the ring fusions (the carbons that join each set of two rings together) as cis or trans to each other.
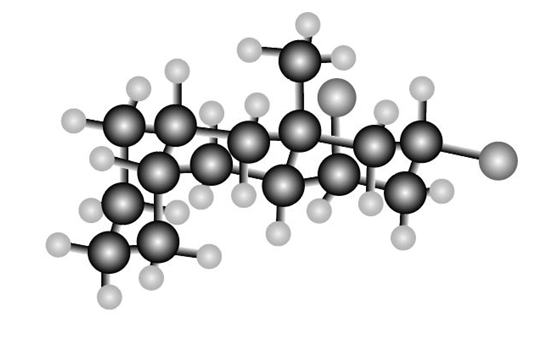
B
What do you think about this solution?
We value your feedback to improve our textbook solutions.