Chapter 4: Q.42. (page 128)
Question: Give the IUPAC name for each compound.
Short Answer
Answer
- 4-isopropylheptane
- 3-ethyl-3-methylpentane
- 4,4-diethyl-5-methyloctane
Learning Materials
Features
Discover
Chapter 4: Q.42. (page 128)
Question: Give the IUPAC name for each compound.
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
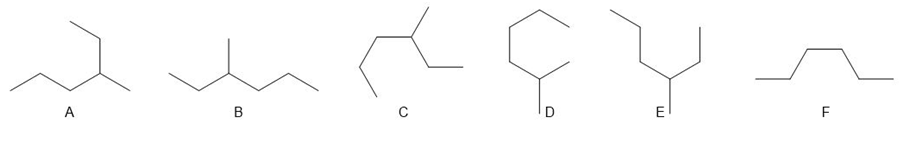
Question: a. Which compounds (B–F) are identical to A? b. Which compounds (B–F) represent an isomer of A?
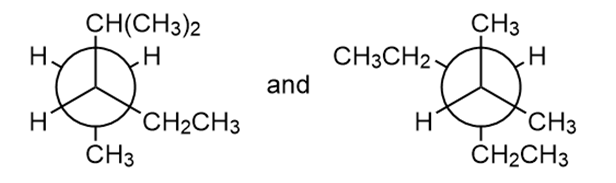
Question: The eclipsed conformation of is role="math" localid="1648620042367" less stable than the staggered conformation. How much is the H, Cl eclipsing interaction worth in destabilization?
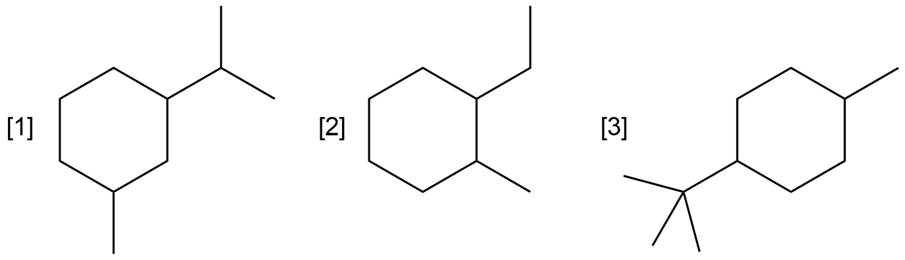
Question: For each compound drawn below:
a. Draw representations for the cis and trans isomers using a hexagon for the six membered ring, and wedges and dashed wedges for substituents.
b. Draw the two possible chair conformations for the cis isomer. Which conformation, if either, is more stable?
c. Draw the two possible chair conformations for the trans isomer. Which conformation, if either, is more stable?
d. Which isomer, cis or trans, is more stable and why?
Question: Cyclopropane and cyclobutane have similar strain energy despite the fact that the C-C-C bond angles of cyclopropane are much smaller than those of cyclobutane. Suggest an explanation for this observation, considering all sources of strain discussed in Chapter 4.
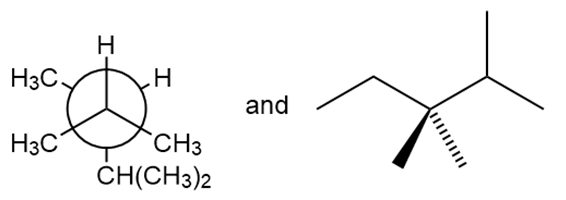
Question: Classify each pair of compounds as constitutional isomers or identical molecules.
a.
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.