Chapter 18: Reactions of Aromatic Compounds
11 P
Intramolecular reactions are also observed in Friedel–Crafts alkylation. Draw the intramolecular alkylation product formed from each of the following reactants. (Watch out for rearrangements!)
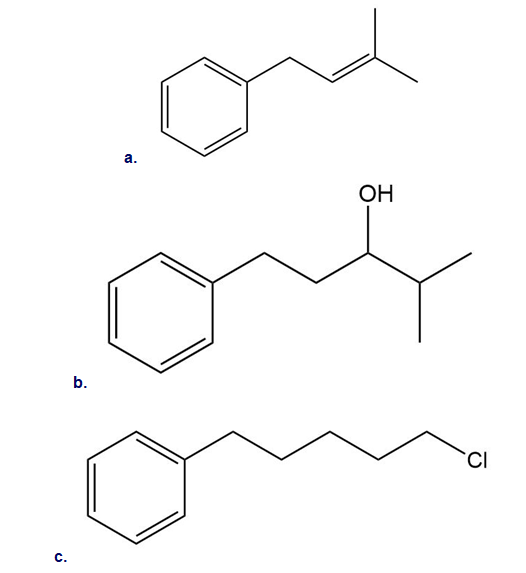
13 P
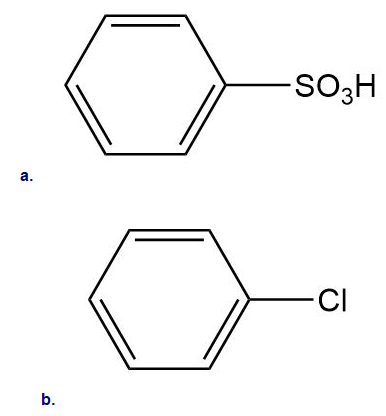
Which substituents have an electron-withdrawing and which have an electron-donating inductive effect
a
b
c
14 P
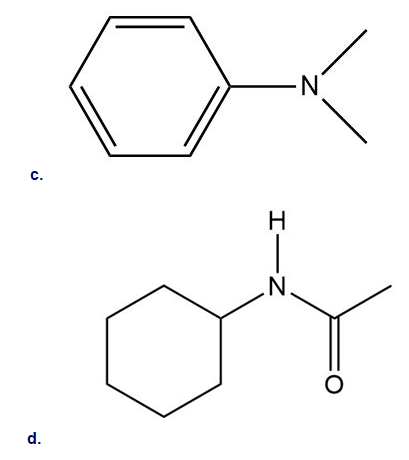
Draw all resonance structures for each compound and use the resonance structures to determine if the substituent has an electron-donating or electron-withdrawing resonance effect.
15 P
Classify each substituent as electron donating or electron withdrawing.
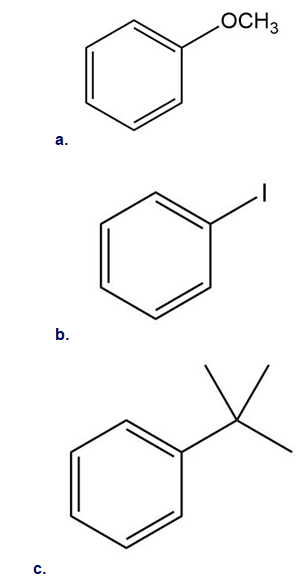
16 P
Draw the products formed when each compound is treated with and . State whether the reaction occurs faster or slower than a similar reaction with benzene.
18 P
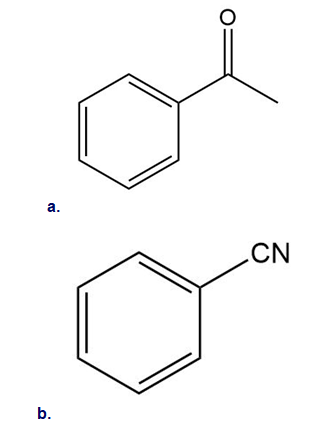
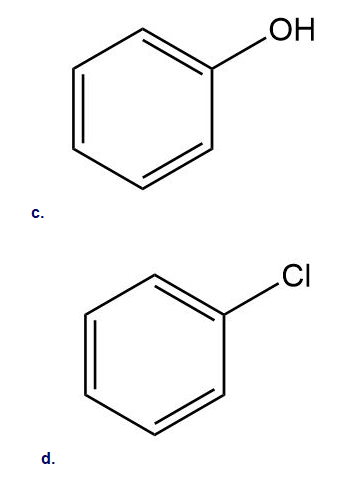
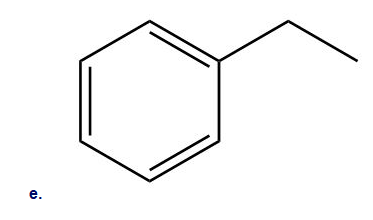
Rank the following compounds in order of increasing reactivity in electrophilic aromatic substitution.
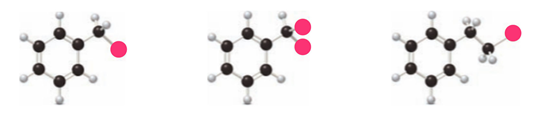
19 P
Draw all resonance structures for the carbocation formed by ortho attack of the electrophile on each starting material. Label any resonance structures that are especially stable or unstable.
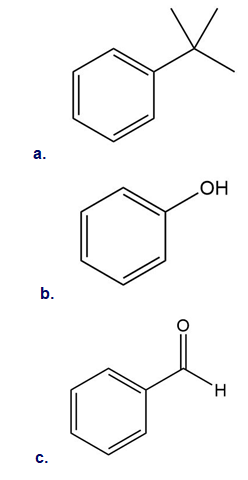
1P
Why is benzene less reactive toward electrophiles than an alkene, even though it has more π electrons than an alkene (six versus two)?
20 P
Draw the products of each reaction.
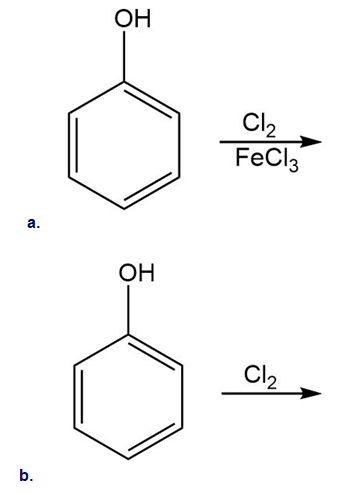
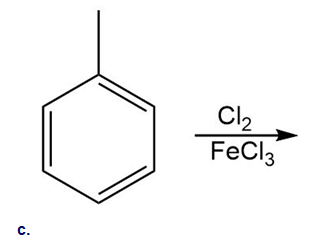
21 P
Which of the following compounds undergo Friedel–Crafts alkylation with and ? Draw the products formed when a reaction occurs.