Chapter 18: 11 P (page 690)
Intramolecular reactions are also observed in Friedel–Crafts alkylation. Draw the intramolecular alkylation product formed from each of the following reactants. (Watch out for rearrangements!)
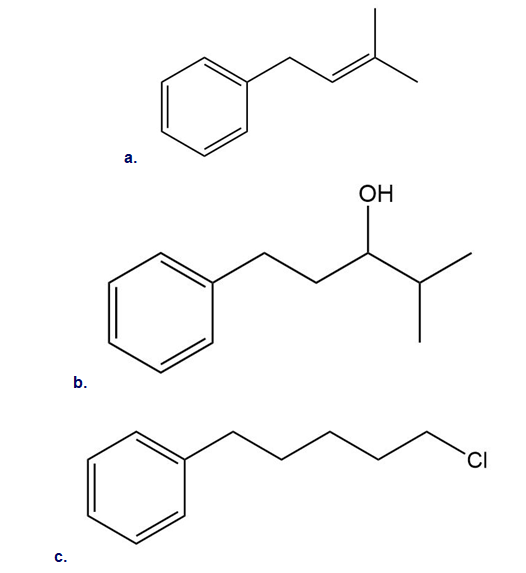
Short Answer
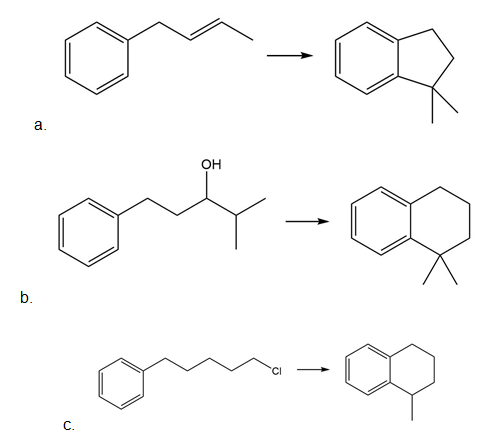
Learning Materials
Features
Discover
Chapter 18: 11 P (page 690)
Intramolecular reactions are also observed in Friedel–Crafts alkylation. Draw the intramolecular alkylation product formed from each of the following reactants. (Watch out for rearrangements!)


All the tools & learning materials you need for study success - in one app.
Get started for free
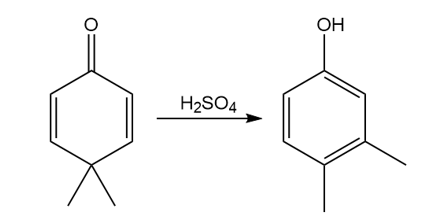
Question: Draw a stepwise, detailed mechanism for the dienone–phenol rearrangement, a reaction that forms alkyl-substituted phenols from cyclohexadienones.
Why is benzene less reactive toward electrophiles than an alkene, even though it has more π electrons than an alkene (six versus two)?
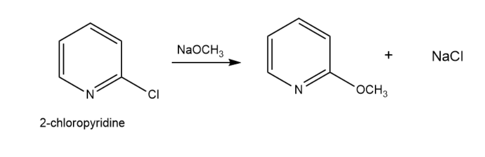
Question: Draw a stepwise mechanism for the following substitution. Explain why 2-chloropyridine reacts faster than chlorobenzene in this type of reaction.
Question: Explain the reactivity and orientation effects observed in each heterocycle.
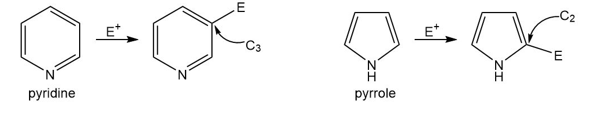
a. Pyridine is less reactive than benzene in electrophilic aromatic substitution and yields 3-substituted products.
b. Pyrrole is more reactive than benzene in electrophilic aromatic substitution and yields 2-substituted products.
Draw all products formed when m-chlorotoluene is treated with in
What do you think about this solution?
We value your feedback to improve our textbook solutions.