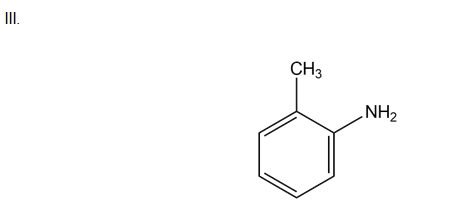
Chapter 18: 27 P (page 709)
Draw all products formed when m-chlorotoluene is treated with in
Short Answer
Three possible products:

Learning Materials
Features
Discover
Chapter 18: 27 P (page 709)
Draw all products formed when m-chlorotoluene is treated with in
Three possible products:


All the tools & learning materials you need for study success - in one app.
Get started for free
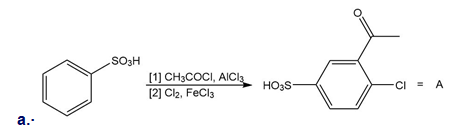
Explain why each of the following reactions will not form the given product. Then, design a synthesis of A from benzene and B from phenol .

Question: Explain the reactivity and orientation effects observed in each heterocycle.

a. Pyridine is less reactive than benzene in electrophilic aromatic substitution and yields 3-substituted products.
b. Pyrrole is more reactive than benzene in electrophilic aromatic substitution and yields 2-substituted products.
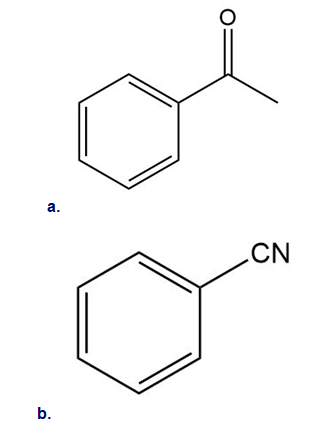
Classify each substituent as electron donating or electron withdrawing.
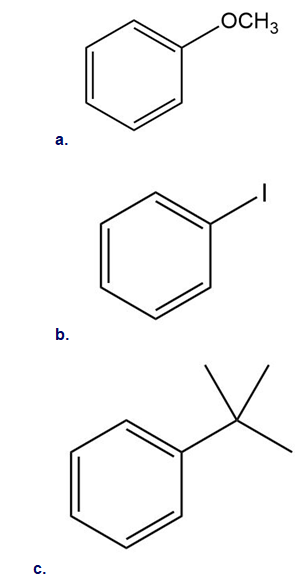
Draw the products formed when each compound is treated with and . State whether the reaction occurs faster or slower than a similar reaction with benzene.


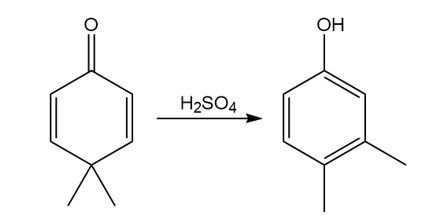
Question: Draw a stepwise, detailed mechanism for the dienone–phenol rearrangement, a reaction that forms alkyl-substituted phenols from cyclohexadienones.
What do you think about this solution?
We value your feedback to improve our textbook solutions.