Chapter 18: 29 P (page 711)
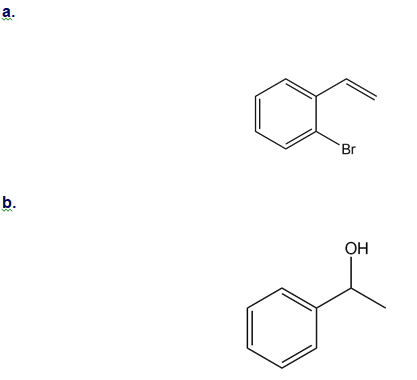
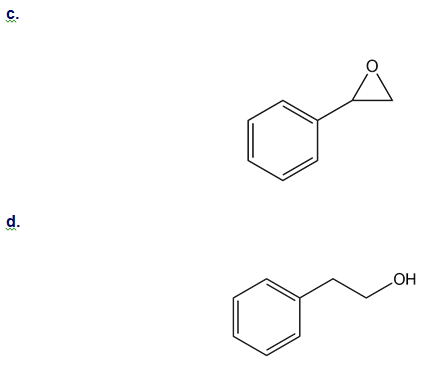
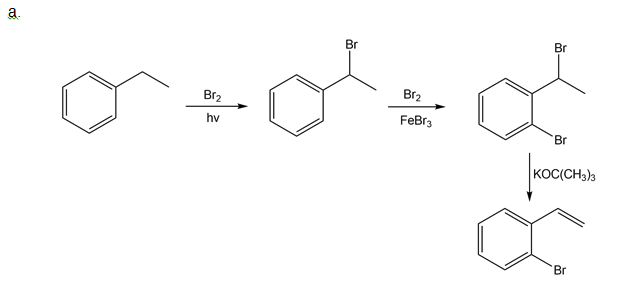
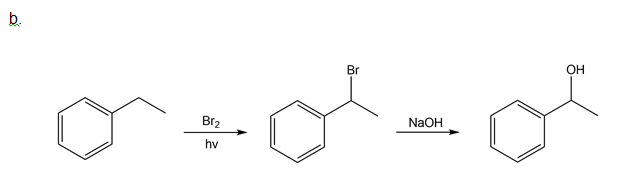
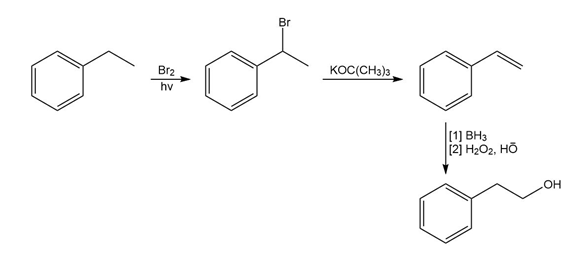
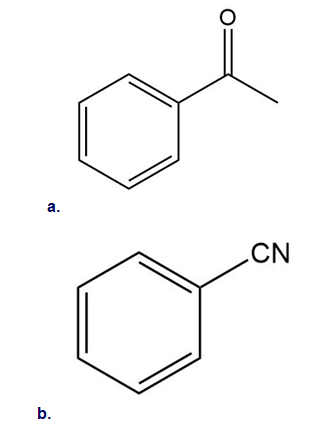
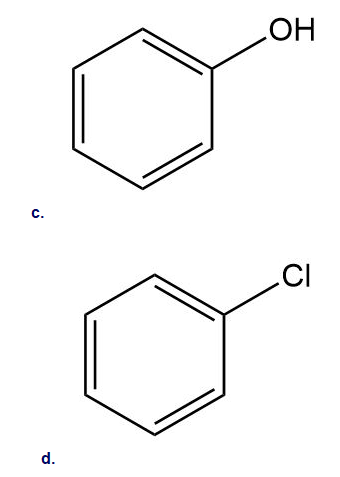
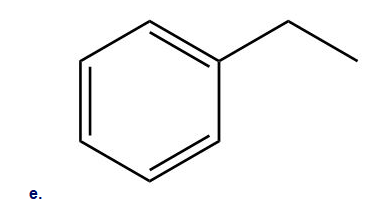
How could you use ethylbenzene to prepare each compound? More than one step is required.
Short Answer
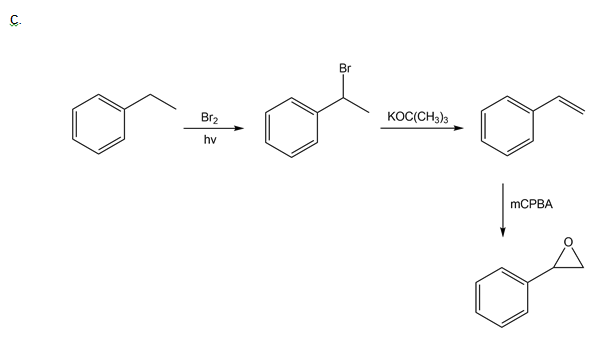
d)
Learning Materials
Features
Discover
Chapter 18: 29 P (page 711)
How could you use ethylbenzene to prepare each compound? More than one step is required.





d)

All the tools & learning materials you need for study success - in one app.
Get started for free
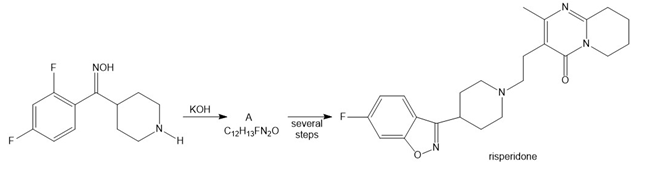
Draw the structure of A, an intermediate in the synthesis of the antipsychotic drug risperidone. Explain why three rings in risperidone are considered aromatic.
Question: Compound X (molecular formula ) was treated with, , to yield compound Y (molecular formula ). Based on theNMR spectra of X and Y were given below, what are the structures of X and Y?
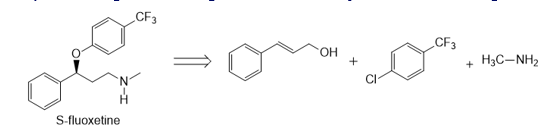
Question: Devise a synthesis of optically active (S)-fluoxetine (trade name Prozac) from the given starting materials and any other needed reagents.
Draw the products formed when each compound is treated with and . State whether the reaction occurs faster or slower than a similar reaction with benzene.
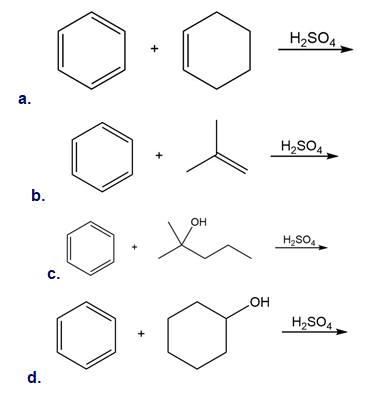
Draw the product of each reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.