Chapter 18: 15 P (page 694)
Classify each substituent as electron donating or electron withdrawing.
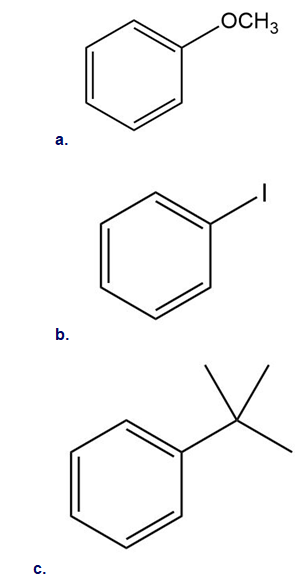
Short Answer
a. Electron donating substituents.
b. Electron withdrawing substituents.
c. Electron donating substituents.
Learning Materials
Features
Discover
Chapter 18: 15 P (page 694)
Classify each substituent as electron donating or electron withdrawing.

a. Electron donating substituents.
b. Electron withdrawing substituents.
c. Electron donating substituents.
All the tools & learning materials you need for study success - in one app.
Get started for free
Why is benzene less reactive toward electrophiles than an alkene, even though it has more π electrons than an alkene (six versus two)?
What products are formed when benzene is treated with each alkyl chloride and ?
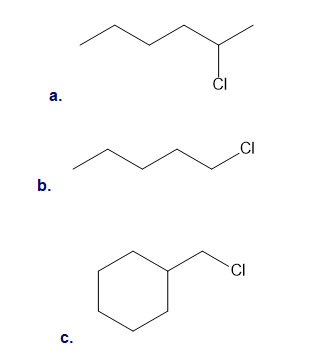
Draw a detailed mechanism for the chlorination of benzene using and .
What is the major product formed by an intramolecular Friedel–Crafts acylation of the following compound?
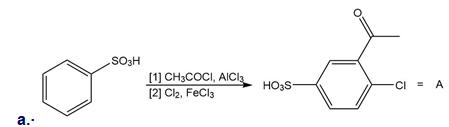
Explain why each of the following reactions will not form the given product. Then, design a synthesis of A from benzene and B from phenol .
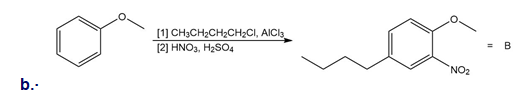
What do you think about this solution?
We value your feedback to improve our textbook solutions.