Chapter 18: 3P (page 682)
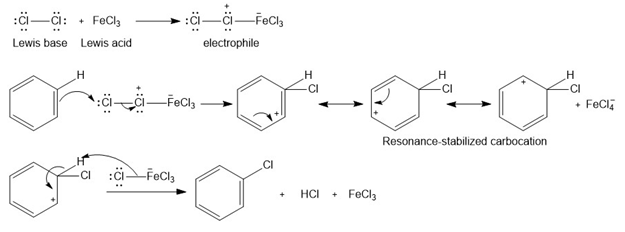
Draw a detailed mechanism for the chlorination of benzene using and .
Short Answer
Learning Materials
Features
Discover
Chapter 18: 3P (page 682)
Draw a detailed mechanism for the chlorination of benzene using and .

All the tools & learning materials you need for study success - in one app.
Get started for free
What is the major product formed by an intramolecular Friedel–Crafts acylation of the following compound?
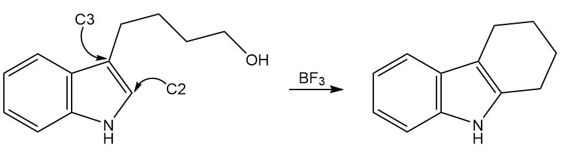
Question: Devise a stepwise mechanism for the following reaction. The reaction does not take place by direct electrophilic aromatic substitution at C2 . (Hint: The mechanism begins with addition of an electrophile at C3.)
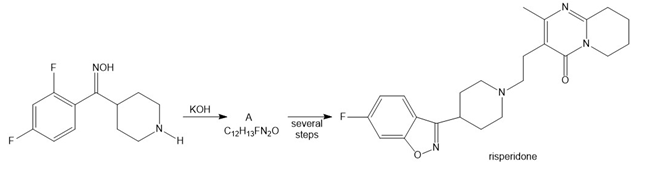
Draw the structure of A, an intermediate in the synthesis of the antipsychotic drug risperidone. Explain why three rings in risperidone are considered aromatic.
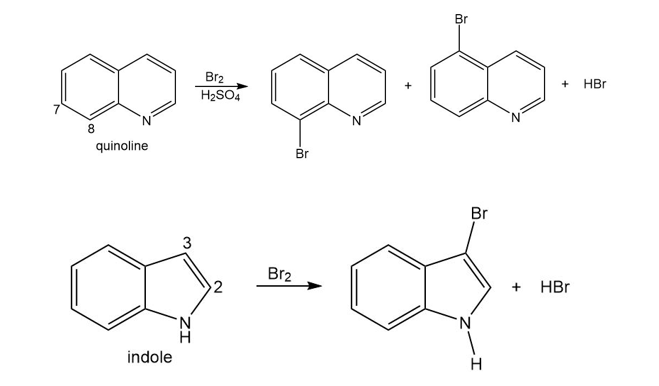
Question: The bicyclic heterocycles quinoline and indole undergo electrophilic aromatic substitution to give the products shown.
a. Explain why electrophilic substitution occurs on the ring without the N atom for quinoline, but occurs on the ring with the N atom in indole.
b. Explain why electrophilic substitution occurs more readily at C8 than C7 in quinoline.
c. Explain why electrophilic substitution occurs more readily at C3 rather than C2 of indole.
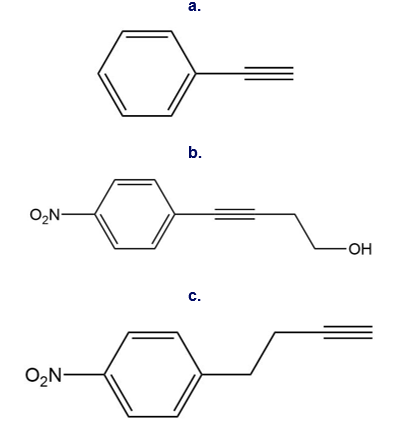
Question: Use the reactions in this chapter along with those learned in Chapters 11 and 12 to synthesize each compound. You may use benzene, acetylene, two-carbon alcohols, ethylene oxide and any organic reagents.
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.