Chapter 18: 40 P (page 722)
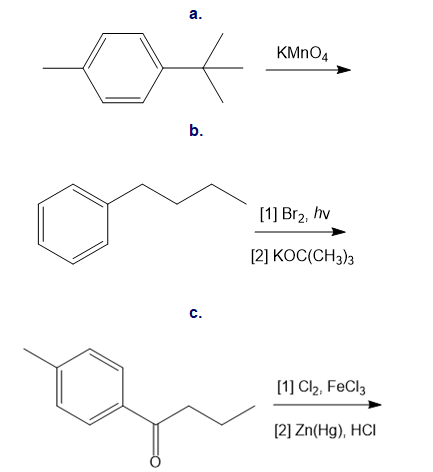
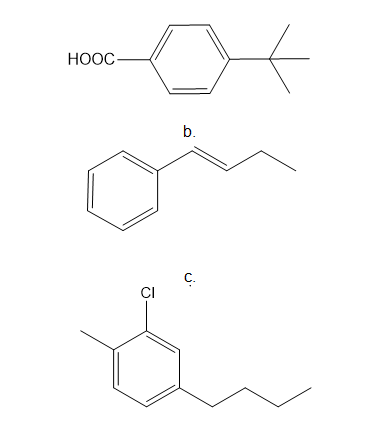
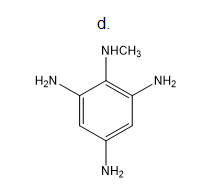
Draw the products of each reaction.
Short Answer
a.
Learning Materials
Features
Discover
Chapter 18: 40 P (page 722)
Draw the products of each reaction.


a.


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Explain the reactivity and orientation effects observed in each heterocycle.
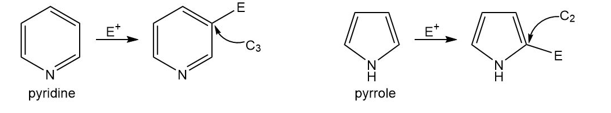
a. Pyridine is less reactive than benzene in electrophilic aromatic substitution and yields 3-substituted products.
b. Pyrrole is more reactive than benzene in electrophilic aromatic substitution and yields 2-substituted products.
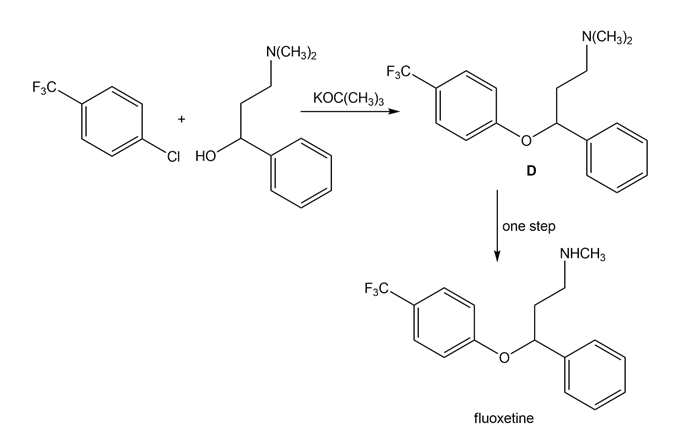
Draw a stepwise mechanism for the following reaction that forms ether D. D can be converted to the antidepressant fluoxetine (trade name Prozac) in a single step.
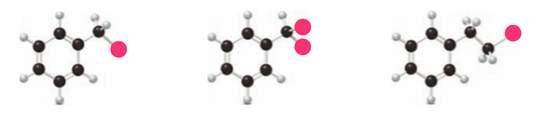
Rank the following compounds in order of increasing reactivity in electrophilic aromatic substitution.
Question: Ibufenac, a para-disubstituted arene with the structure HO2CCH2C6H4CH2CH(CH3)2, is a much more potent analgesic than aspirin, but it was never sold commercially because it caused liver toxicity in some clinical trials. Devise a synthesis of ibufenac from benzene and organic halides having fewer than five carbons.
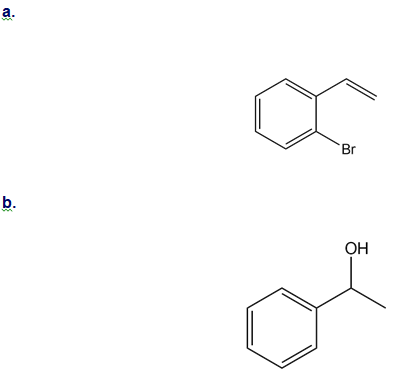
How could you use ethylbenzene to prepare each compound? More than one step is required.
What do you think about this solution?
We value your feedback to improve our textbook solutions.