Chapter 18: 18 P (page 697)
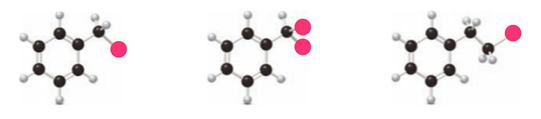
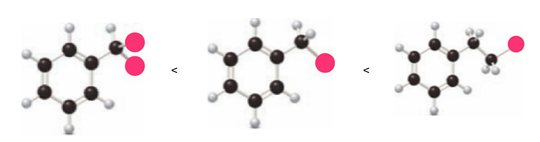
Rank the following compounds in order of increasing reactivity in electrophilic aromatic substitution.
Short Answer
Learning Materials
Features
Discover
Chapter 18: 18 P (page 697)
Rank the following compounds in order of increasing reactivity in electrophilic aromatic substitution.


All the tools & learning materials you need for study success - in one app.
Get started for free
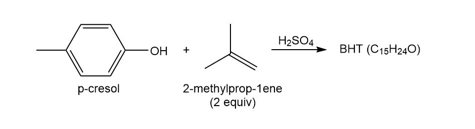
Question: Reaction of p-cresol with two equivalents of 2-methylprop-1-ene affords BHT, a preservative with molecular formula . BHT gives the following NMR spectral data: 1.4 (singlet, 18 H), 2.27 (singlet, 3 H), 5.0 (singlet, 1 H), and 7.0 (singlet, 2 H) ppm. What is the structure of BHT? Draw a stepwise mechanism illustrating how it is formed
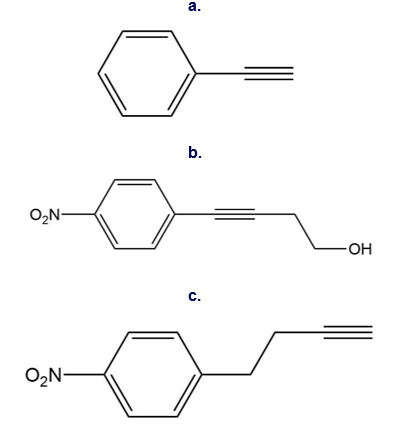
Question: Use the reactions in this chapter along with those learned in Chapters 11 and 12 to synthesize each compound. You may use benzene, acetylene, two-carbon alcohols, ethylene oxide and any organic reagents.
d.
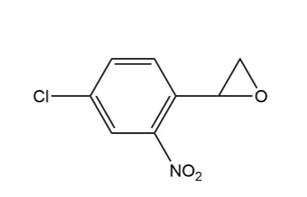
Draw the products of each reaction.
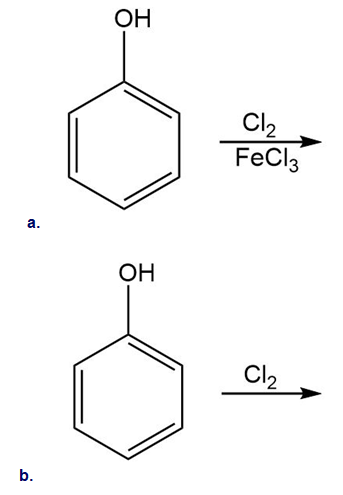
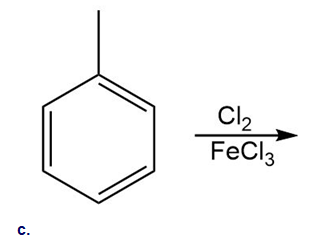
Draw a detailed mechanism for the chlorination of benzene using and .
Write out the two-step sequence that converts benzene to each compound.
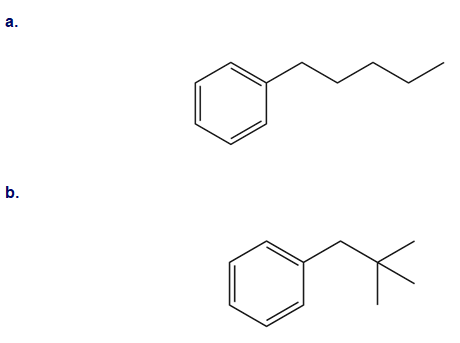
What do you think about this solution?
We value your feedback to improve our textbook solutions.