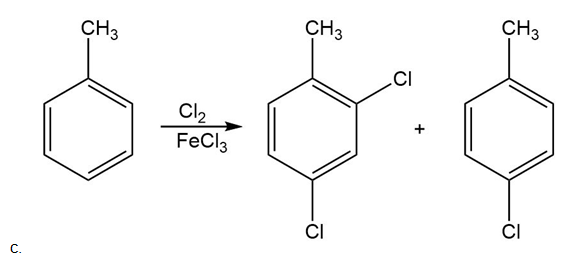
Chapter 18: 20 P (page 702)
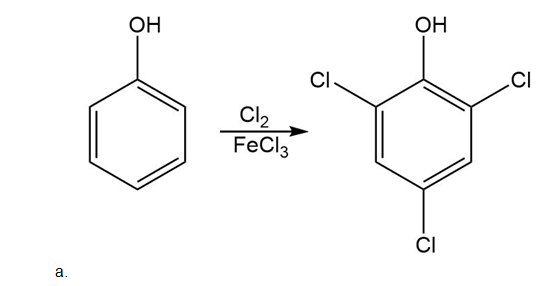
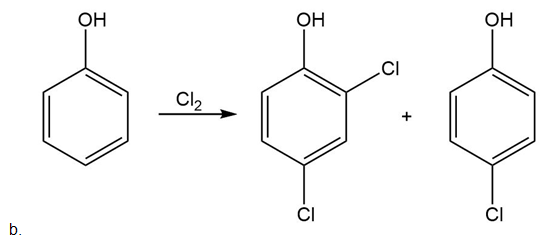
Draw the products of each reaction.
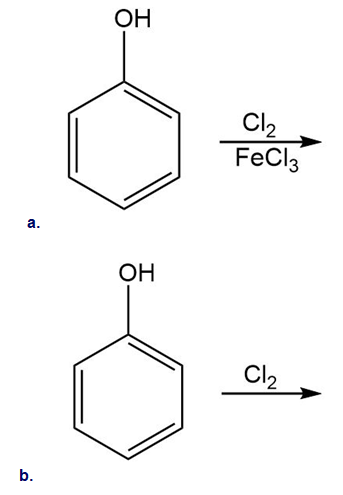
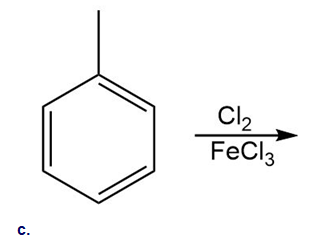
Short Answer
Learning Materials
Features
Discover
Chapter 18: 20 P (page 702)
Draw the products of each reaction.





All the tools & learning materials you need for study success - in one app.
Get started for free
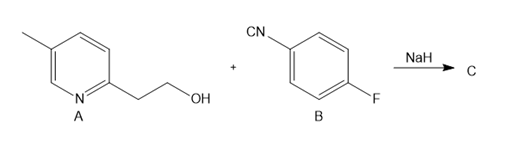
One step in the synthesis of pioglitazone (trade name Actos), a drug used to treat type 2 diabetes, involves the reaction of A with B in the presence of NaH to afford C. What is the structure of C?
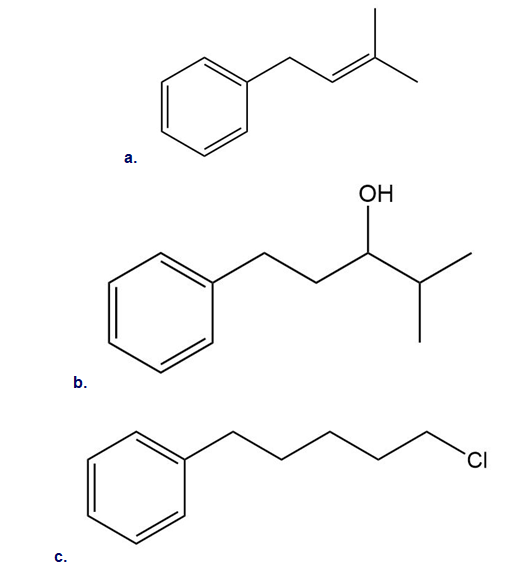
Intramolecular reactions are also observed in Friedel–Crafts alkylation. Draw the intramolecular alkylation product formed from each of the following reactants. (Watch out for rearrangements!)
Question: Explain the reactivity and orientation effects observed in each heterocycle.
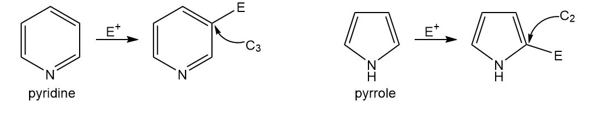
a. Pyridine is less reactive than benzene in electrophilic aromatic substitution and yields 3-substituted products.
b. Pyrrole is more reactive than benzene in electrophilic aromatic substitution and yields 2-substituted products.
Draw the products of each reaction
a)
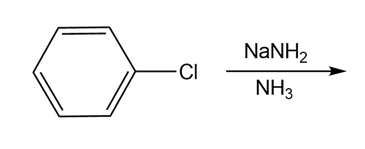
b)
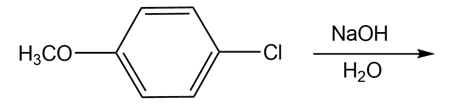
c)
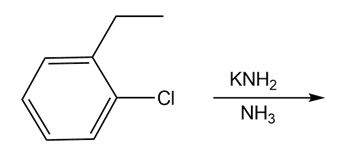
What product is formed when benzene is treated with each organic halide in the presence of .
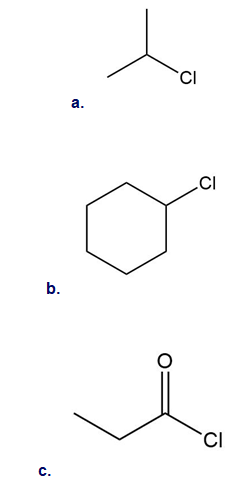
What do you think about this solution?
We value your feedback to improve our textbook solutions.