Chapter 18: 21 P (page 702)
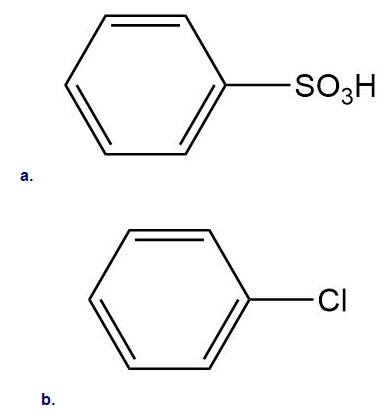
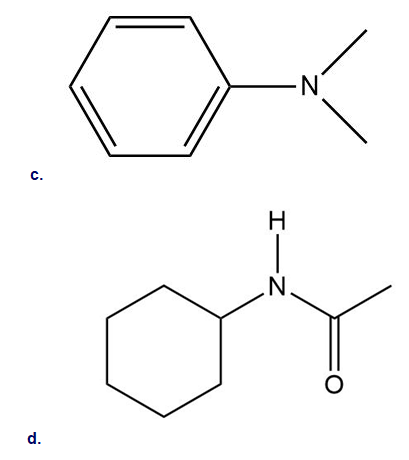
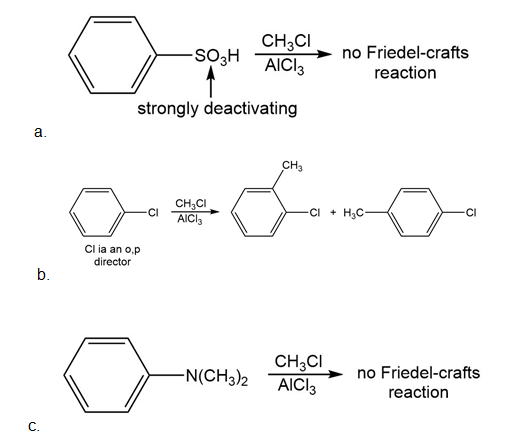
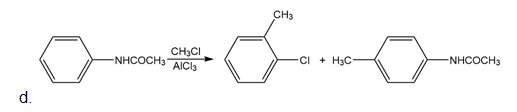
Which of the following compounds undergo Friedel–Crafts alkylation with and ? Draw the products formed when a reaction occurs.
Short Answer
Learning Materials
Features
Discover
Chapter 18: 21 P (page 702)
Which of the following compounds undergo Friedel–Crafts alkylation with and ? Draw the products formed when a reaction occurs.




All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Using resonance structures, explain why a nitroso group (-NO) is an ortho, para director that deactivates a benzene ring toward electrophilic attack.
Draw the products formed when phenol ( ) is treated with each set of reagents.
a. [1] , ; [2] Sn, HCl
b. [1] ; [2] Zn(Hg), HCl
c. [1] ; [2]
d. [1] ; [2]
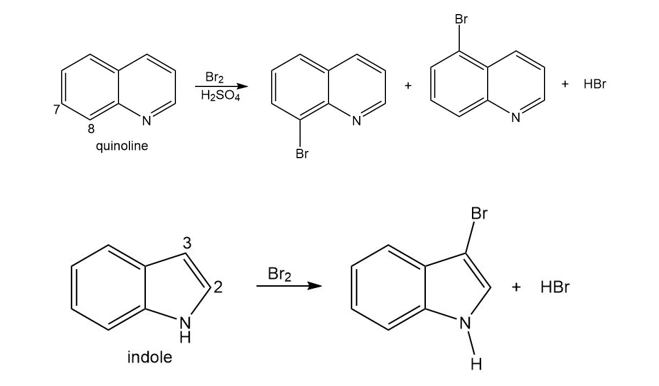
Question: The bicyclic heterocycles quinoline and indole undergo electrophilic aromatic substitution to give the products shown.
a. Explain why electrophilic substitution occurs on the ring without the N atom for quinoline, but occurs on the ring with the N atom in indole.
b. Explain why electrophilic substitution occurs more readily at C8 than C7 in quinoline.
c. Explain why electrophilic substitution occurs more readily at C3 rather than C2 of indole.
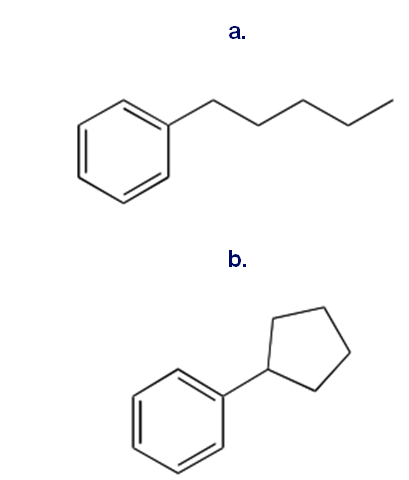
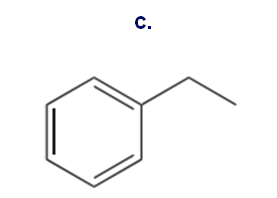
You have learned two ways to make an alkyl benzene: Friedel–Crafts alkylation, and Friedel–Crafts acylation followed by reduction. Although some alkyl benzenes can be prepared by both methods, it is often true that only one method can be used to prepare a given alkyl benzene. Which method(s) can be used to prepare each of the following compounds from benzene? Show the steps that would be used.
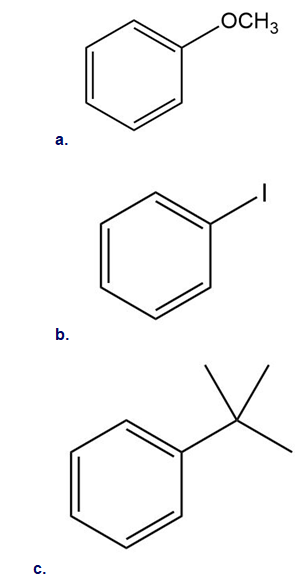
Classify each substituent as electron donating or electron withdrawing.
What do you think about this solution?
We value your feedback to improve our textbook solutions.