Chapter 18: Q 51. (page 723)
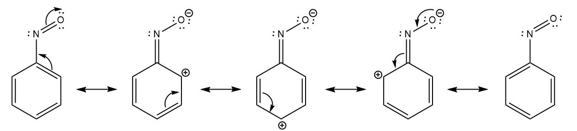
Question: Using resonance structures, explain why a nitroso group (-NO) is an ortho, para director that deactivates a benzene ring toward electrophilic attack.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 18: Q 51. (page 723)
Question: Using resonance structures, explain why a nitroso group (-NO) is an ortho, para director that deactivates a benzene ring toward electrophilic attack.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
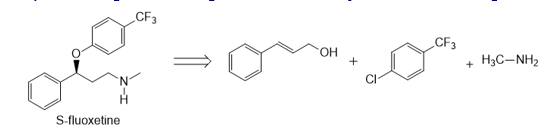
Question: Devise a synthesis of optically active (S)-fluoxetine (trade name Prozac) from the given starting materials and any other needed reagents.
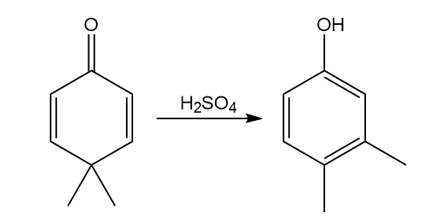
Question: Draw a stepwise, detailed mechanism for the dienone–phenol rearrangement, a reaction that forms alkyl-substituted phenols from cyclohexadienones.
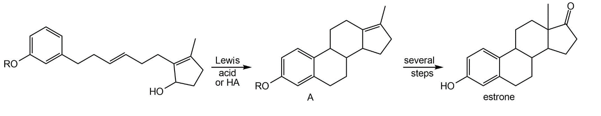
Question: Draw a stepwise mechanism for the following intramolecular reaction, which is used in the synthesis of the female sex hormone estrone.
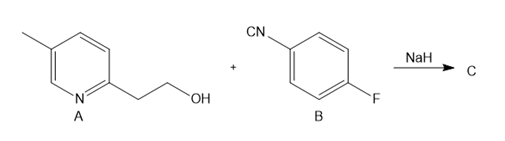
One step in the synthesis of pioglitazone (trade name Actos), a drug used to treat type 2 diabetes, involves the reaction of A with B in the presence of NaH to afford C. What is the structure of C?
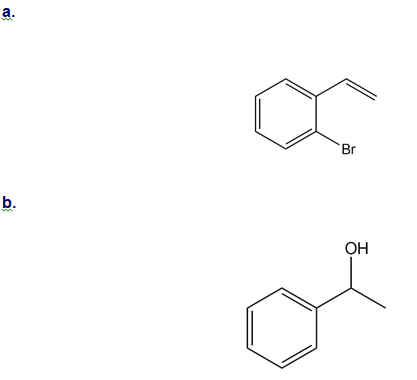
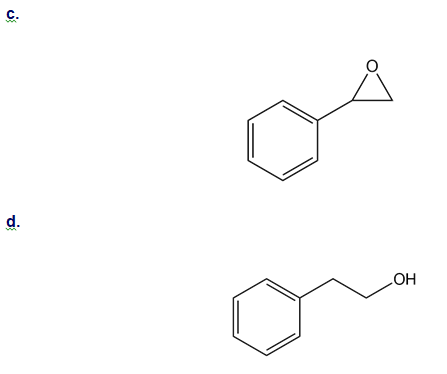
How could you use ethylbenzene to prepare each compound? More than one step is required.
What do you think about this solution?
We value your feedback to improve our textbook solutions.