Chapter 18: Q 76. (page 728)
Question: Draw a stepwise, detailed mechanism for the dienone–phenol rearrangement, a reaction that forms alkyl-substituted phenols from cyclohexadienones.
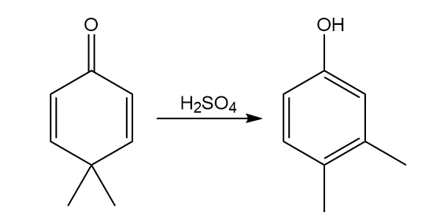
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 18: Q 76. (page 728)
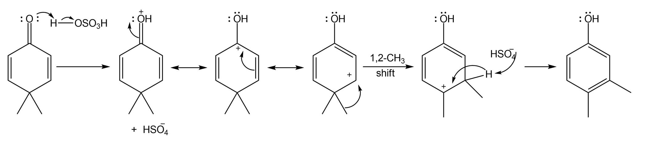
Question: Draw a stepwise, detailed mechanism for the dienone–phenol rearrangement, a reaction that forms alkyl-substituted phenols from cyclohexadienones.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
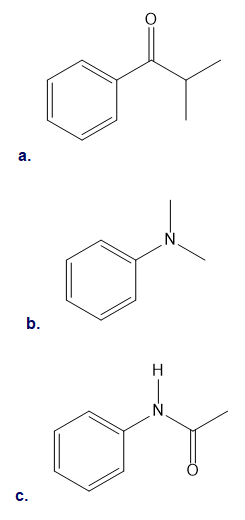
Draw the products formed when each compound is treated with
In Step [2] of Mechanism 18.1, loss of a proton to form the substitution product was drawn using one resonance structure only. Use curved arrows to show how the other two resonance structures can be converted to the substitution product (PhE) by removal of a proton with: B.
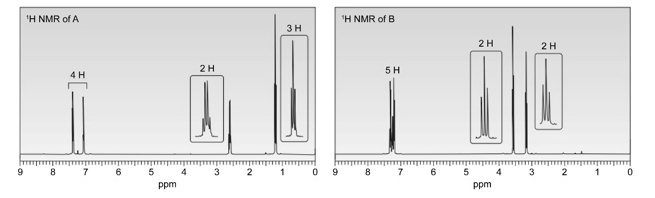
Question: Identify the structures of isomers A and B (molecular formula )
Draw the products of each reaction
a)
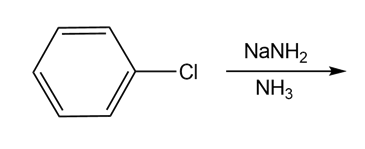
b)
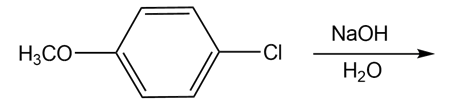
c)
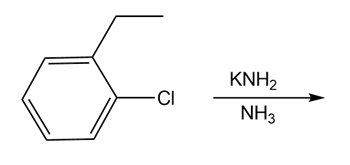
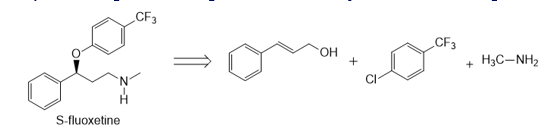
Question: Devise a synthesis of optically active (S)-fluoxetine (trade name Prozac) from the given starting materials and any other needed reagents.
What do you think about this solution?
We value your feedback to improve our textbook solutions.