Chapter 18: Q 69. (page 726)
Question: Identify the structures of isomers A and B (molecular formula )

Short Answer
Answer
Structure of isomer A
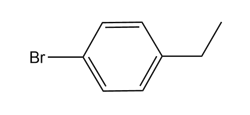
Structure of isomer B

Learning Materials
Features
Discover
Chapter 18: Q 69. (page 726)
Question: Identify the structures of isomers A and B (molecular formula )

Answer
Structure of isomer A

Structure of isomer B

All the tools & learning materials you need for study success - in one app.
Get started for free
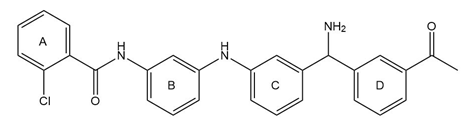
Consider the tetracyclic aromatic compound drawn below, with rings labeled as A, B, C, and D. (a) Which of the four rings is most reactive in electrophilic aromatic substitution? (b) Which of the four rings is least reactive in electrophilic aromatic substitution? (c) What are the major product(s) formed when this compound is treated with one equivalent of ?
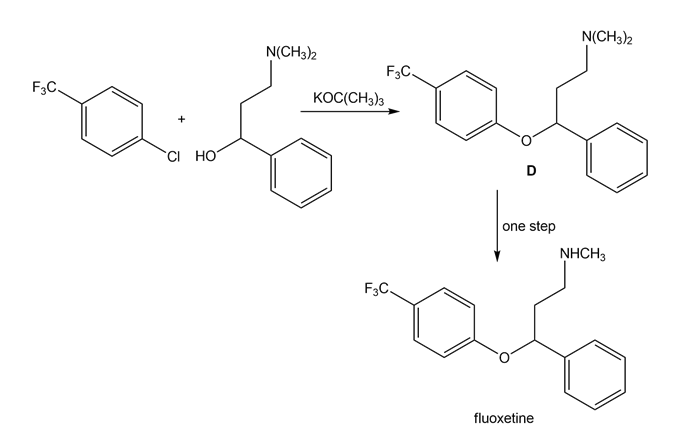
Draw a stepwise mechanism for the following reaction that forms ether D. D can be converted to the antidepressant fluoxetine (trade name Prozac) in a single step.
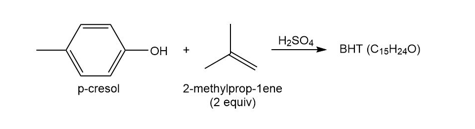
Question: Reaction of p-cresol with two equivalents of 2-methylprop-1-ene affords BHT, a preservative with molecular formula . BHT gives the following NMR spectral data: 1.4 (singlet, 18 H), 2.27 (singlet, 3 H), 5.0 (singlet, 1 H), and 7.0 (singlet, 2 H) ppm. What is the structure of BHT? Draw a stepwise mechanism illustrating how it is formed
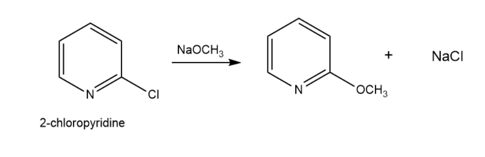
Question: Draw a stepwise mechanism for the following substitution. Explain why 2-chloropyridine reacts faster than chlorobenzene in this type of reaction.
Why is benzene less reactive toward electrophiles than an alkene, even though it has more π electrons than an alkene (six versus two)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.