Chapter 18: 19 P (page 700)
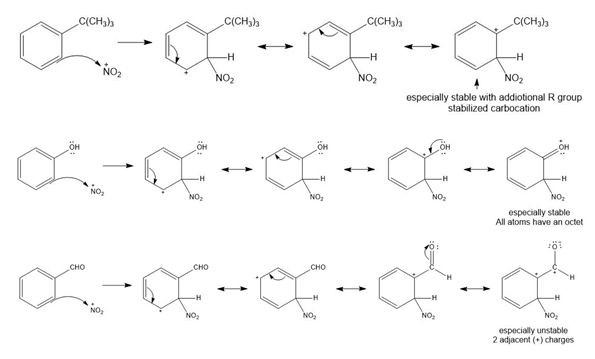
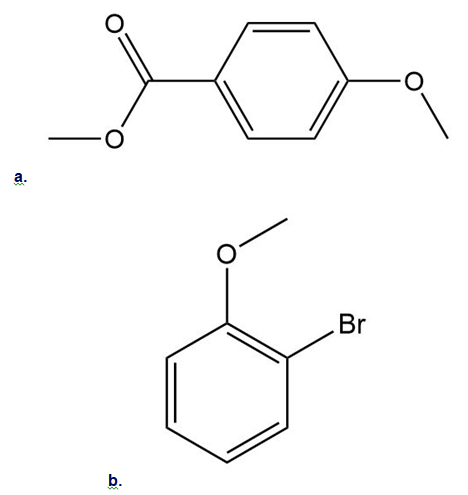
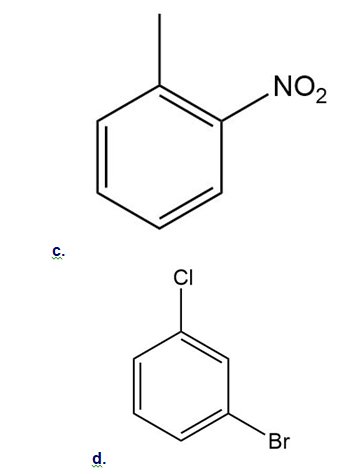
Draw all resonance structures for the carbocation formed by ortho attack of the electrophile on each starting material. Label any resonance structures that are especially stable or unstable.
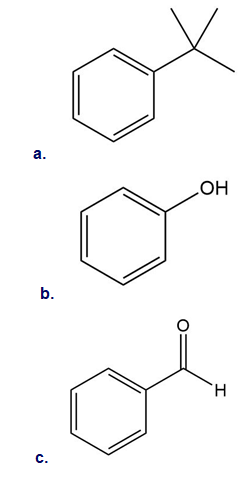
Short Answer
Learning Materials
Features
Discover
Chapter 18: 19 P (page 700)
Draw all resonance structures for the carbocation formed by ortho attack of the electrophile on each starting material. Label any resonance structures that are especially stable or unstable.


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Devise a stepwise mechanism for the following reaction. The reaction does not take place by direct electrophilic aromatic substitution at C2 . (Hint: The mechanism begins with addition of an electrophile at C3.)
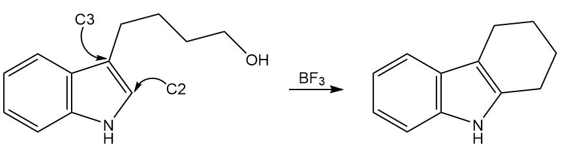
Question: Using resonance structures, explain why a nitroso group (-NO) is an ortho, para director that deactivates a benzene ring toward electrophilic attack.
Rank the compounds in each group in order of increasing reactivity in electrophilic aromatic substitution: (a) ; (b) .
For each N-substituted benzene, predict whether the compound reacts faster than, slower than, or at a similar rate to benzene in electrophilic aromatic substitution. Then draw the major product (s) formed when each compound reacts with a general electrophile .
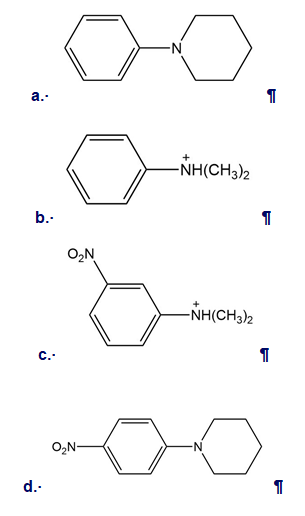
Draw the products formed when each compound is treated with and .
What do you think about this solution?
We value your feedback to improve our textbook solutions.