Chapter 18: 13 P (page 691)
Which substituents have an electron-withdrawing and which have an electron-donating inductive effect
a
b
c
Short Answer
a. Electron-donating group.
b. Electron-withdrawing group.
c. Electron-withdrawing group.
Learning Materials
Features
Discover
Chapter 18: 13 P (page 691)
Which substituents have an electron-withdrawing and which have an electron-donating inductive effect
a
b
c
a. Electron-donating group.
b. Electron-withdrawing group.
c. Electron-withdrawing group.
All the tools & learning materials you need for study success - in one app.
Get started for free
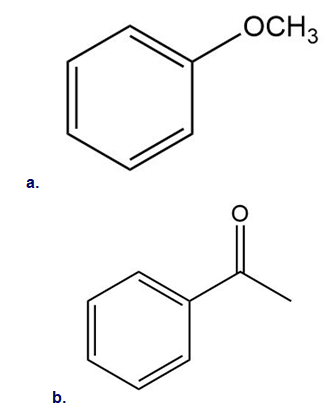
Draw all products formed when m-chlorotoluene is treated with in
Draw all resonance structures for each compound and use the resonance structures to determine if the substituent has an electron-donating or electron-withdrawing resonance effect.
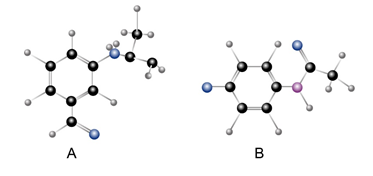
Draw the products formed when A and B are treated with each of the following reagents:
(a),; (b)l3; (c)
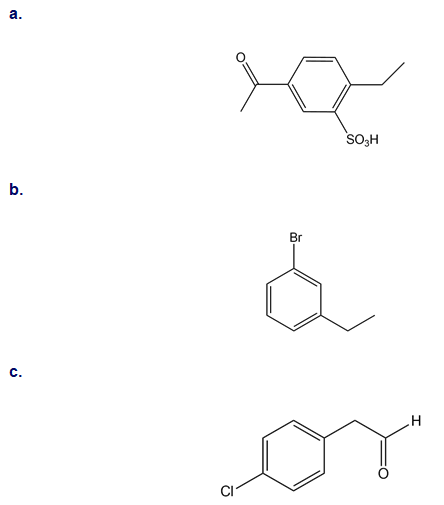
Synthesize each compound from benzene
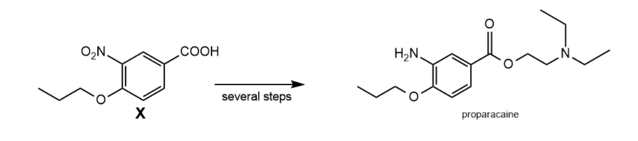
Question: Carboxylic acid X is an intermediate in the multistep synthesis of proparacaine, a local anesthetic. Devise a synthesis of X from phenol and any needed organic or inorganic reagents.
What do you think about this solution?
We value your feedback to improve our textbook solutions.