Chapter 19: Q. 2 (page 543)
How much work is done on the gas in the process shown in Figure EX19.2?
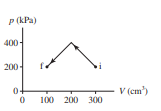
Short Answer
The amount of work done energy is 60J
Learning Materials
Features
Discover
Chapter 19: Q. 2 (page 543)
How much work is done on the gas in the process shown in Figure EX19.2?

The amount of work done energy is 60J
All the tools & learning materials you need for study success - in one app.
Get started for free
A 65 cm3 block of iron is removed from an 800C furnace and
immediately dropped into 200 mL of 20C water. What fraction
of the water boils away?
a. 100 J of heat energy are transferred to 20 g of mercury. By how much does the temperature increase?
b. How much heat is needed to raise the temperature of 20 g of water by the same amount?
A 750 g aluminum pan is removed from the stove and plunged
into a sink filled with 10.0 L of water at 20.0 C. The water temperature
quickly rises to 24.0 C. What was the initial temperature
of the pan in C and in F?
w
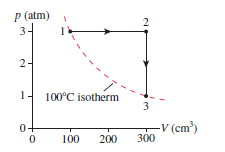
A monatomic gas follows the process 1S 2S 3 shown in
Figure EX19.32. How much heat is needed for (a) process 1S 2
and (b) process 2S 3?
What do you think about this solution?
We value your feedback to improve our textbook solutions.