Chapter 19: Q 32. (page 544)
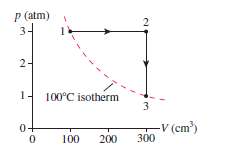
A monatomic gas follows the process 1S 2S 3 shown in
Figure EX19.32. How much heat is needed for (a) process 1S 2
and (b) process 2S 3?
Short Answer
The heat needed for process (a) is 150 J and for process (b) -90 J.
Learning Materials
Features
Discover
Chapter 19: Q 32. (page 544)
A monatomic gas follows the process 1S 2S 3 shown in
Figure EX19.32. How much heat is needed for (a) process 1S 2
and (b) process 2S 3?

The heat needed for process (a) is 150 J and for process (b) -90 J.
All the tools & learning materials you need for study success - in one app.
Get started for free
In Problems 74 through 76 you are given the equation used to solve a problem. For each of these, you are to
a. Write a realistic problem for which this is the correct equation.
b. Finish the solution of the problem.
74.
A 65 cm3 block of iron is removed from an 800C furnace and
immediately dropped into 200 mL of 20C water. What fraction
of the water boils away?
|| A 10 m14 m house is built on a 12-cm-thick concrete slab.
What is the heat-loss rate through the slab if the ground temperature
is 5C while the interior of the house is 22C?
Suppose you take and hold a deep breath on a chilly day, inhaling 3.0 L of air at 0°C and 1 atm. a. How much heat must your body supply to warm the air to your internal body temperature of 37°C? b. By how much does the air’s volume increase as it warms?
The gas cylinder in Figure Q19.10 is well insulated except for the bottom surface, which is in contact with a block of ice.
The piston can slide without friction. The initial gas temperature is
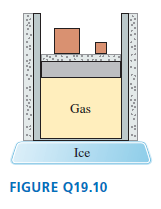
a. During the process that occurs until
the gas reaches a new equilibrium, are
(i) , (ii) W, and (iii) Q greater than, less than, or equal to zero? Explain.
b. Draw a pV diagram showing the process.
What do you think about this solution?
We value your feedback to improve our textbook solutions.